Synthesis of Lignin Monomers
Introduction
Lignin is the second most abundant organic polymer. It is found in plants, majorly in trees. Its function is to serve in the assistance of water flow throughout the plant. It forms a barrier for the evaporation, trapping the water inside.
The structure of lignin is composed of the seemingly random combination of three monomers: p-coumaryl alcohol, coniferyl alcohol, and sinapyl alcohol. The monomers only differ by the number of methoxy groups present on the phenol. The phenol group contributes the the overall stability of the molecules' formed radicals. These radicals determine the bonding spot between monomers when creating lignin. These monomers can also join together to form dimers that are known as Lignan; which has biological activity.
These lignin monomers are derived from their acid precursors through the reduction of an aldehyde to the corresponding alcohol. While the acid precursors,p-courmaric acid (p-coumaryl alcohol), ferulic acid (coniferyl alcohol), and sinapic acid (sinapyl alcohol) may not be as abundant in nature, when it comes to the lab setting, these chemicals are easier to come by than their alcohol forms. The synthesis of theses alcohols can save a research space a great deal of money when experiments require the alcohol forms of these monomers; such as others in the Sturgeon research group.
Materials
Diisobutylaluminum hydride solution (25 wt.% in toluene)
Product Number: 192724-100G
Quantity: 2
Price: $52.20
Toluene (Anhydrous, 99.8%)
Product Number: 244511-1L
Quantity: 1
Price: $60.50
Product Number: 708496-100mL
Quantity: 1
Price: $45.50
Other General Lab Materials
- Ethanol
- Sodium Bicarbonate
- Brine
- Magnesium Sulfate
Lab Hardware
- Various sized Round Bottom Flasks
- Heating Mantle
- Reflux Condenser
- Water Pump
- Separation Funnel
- Rotovap Vacuum Distiller
- Bubbler
- Dririte Air/Gas Dryer
Experimental
The process can most simply be broken up into two main steps
Step One: Esterification (Figure 2)
The esterification of ferulic acid and other monomers was the first step in the reduction of acid to alcohol by replacing the carboxylic acid group with an ethyl ester protecting group. A 0.24M solution of ferulic acid in ethanol (250 mL ethanol, 12.1g ferulic acid) along with 3 drops of concentrated HCl was allowed to reflux for two days. The resulting solution was evaporated at low pressure, leaving a solid. This solid was then resolubilized in ethyl acetate (250 mL) and rinsed with sodium bicarbonate (2x100 mL) and brine (50 mL). The solution was dried using magnesium sulfate and evaporated at low pressure, resulting in an oil like solution. The final product was tested in the HPlc (Figure 1).
A second attempt at the esterification of ferulic acid was carried out using the same method as above, but on a 1/5 scale. (Figure 3) However, this time instead of using 3 drops of concentrated HCL, 5 mL of acetyl chloride was added to the solution before reflux. All other reflux solutions used the protocol containing the acetyl chloride.
A third esterification process was ran using a 0.15M solution of ferulic acid ethanol (250mL ethanol, 8.2g ferulic acid) along with 25mL of acetyl chloride. All other parameters were the same as the initial esterification. (Figure 5)
This process also applies to the other monomoers of lignin. This inital process was run on p-coumaric acid to for p-coumaric acid ethyl ester (Figure 7)
Step Two: DIBAL-H Reduction (Figure 4)
The reduction process was done using the strong reducing agent diisobutylaluminum hydride (DIBAL-H). All processes in step two were done under nitrogen due to the strong reactive properties of DIBAL-H with oxygen and hydrogen. The product from step one (third attempt) was placed into a three-armed round bottom flask containing a stir bar and 100mL of toluene was added via syringe and placed in an ice bath. Approximately 52mL of DIBAL-H was added to the solution through an addition funnel over a 10 minute time span while stirring. The reaction stirred for 1 hour. After 1 hour the solution was quenched with 5mL of ethanol. The solution was then rotovapped at 40 degrees Celsius. After evaporation, 100mL of water was added to the solution. The solution was extracted using 2 rinses of 150 mL ethyl acetate and finally rotovapped until a solid product formed. (Figure 6)
Results
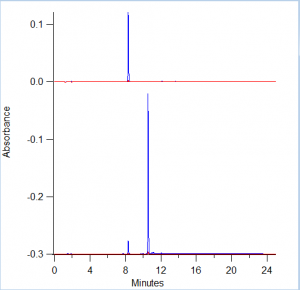
Figure 1 displays the results of HPLC testing that was run on 3 separate samples. The first is the ferulic acid standard. The second is a sample of the final product from the first attempt at the esterfication of ferulic acid. The third is a more dilute sample of the same sample from the second chromatogram. All samples were run using the acquisition method: pCou_100417_SAZT.
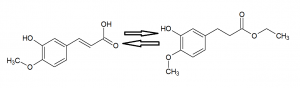
Figure 2 displays the chemical reaction of ferulic acid into ethyl ferulate. This displays the chemical process that occurs during the first steps of this reaction. There is a substitution of functional groups from the carboxylic acid to an ethyl ester protecting group.
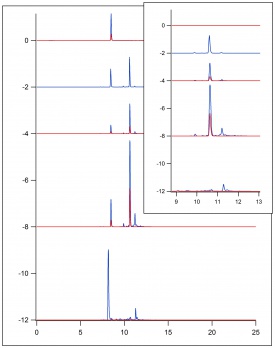
Figure 3 displays the results of HPLC testing that was run on 3 separate samples. The first graph represents the sample of the reflux intermediate from the second attempt at the esterfication process.The second is the ferulic acid standard. The third is the sample of the reflux intermediate from the second attempt at esterfication of ferulic acid after one and a half days of reflux. The fourth graph is the results from a sample of the final product, indicating the esterfication of ferulic acid to ethyl ferulate. The fifth graph is the product, coniferyl alcohol, after the DIABL-H steps of the reaction. All samples were run using the acquisition method: pCou_100417_SAZT.
Figure 4 displays the chemical reaction Ethyl Ferulate to Coniferyl Alcohol. This displays the chemical process that occurs during the second steps of this reaction. The ethyl ester protecting group is reduced to the alcohol functional group.
Figure 5 displays the chromatograms that were produced from running the solution in the HPLC. The initial chromatogram is the Ferulic acid solution before reflux. The Next three are the ethyl ferulate solution after 24 hours of reflux, after 48 hours of reflux, and after the solution had been rotovapped and gone through the extraction process.
Figure 6 displays the chromatograms for the material after the DIBAL-H portion of the reaction. The first is the solution after the initial reduction process. The second is the solution after it had been rotovapped and extracted. The final chromatogram is the coniferyl alcohol standard.
Figure 7 displays chromatograms of HPLC results for a p-coumaric acid standand and then the p-coumaric acid ethyl ester.
Discussion
Definite progress was made in the ability to synthesize the alcohol form of the lignin monomers; there is still a great deal of work to be done. A clear improvement in purity of the samples was seen from the initial trials to the later trials. There is still room for improvement when it comes to the purity levels of the substances. It can be proposed that the quality of the final product will improve with the greater number of trials ran. Another way to approach an improvement in purity would be to run the solutions through the flash chromatography instrument after the extraction process of the esterified solution and prior to the DIBAL-H step of the method.
Through trial and error a seemingly more precise method was used in the second and third trials of the esterification step. There are other potential improvements to the procedure. While it is clear that synthesis is possible, this research contains the possibility of larger scale production once the procedure is more precise.
References
Lignin Biosynthesis and Structure
[Overview from Lignin and Lignans: Advances in Chemistry]
Facile Large-Scale Synthesis of Coniferyl, Sinapyl, and p-Coumaryl Alcohol