Oxidation of Lignin Monomers
You have reached the page dedicated to the research of lignin monomers. This page was created by Stephanie Saey and will be maintained by Zelinda Taylor. Stephanie was a 2018 Biochemistry/Biopsychology graduate and Zelinda is currently a Junior Biochemistry research student.
Lignin monomers
Abstract
The biosynthesis of the lignin polymer occurs through oxidative coupling between three basic monomers: p-coumaryl alcohol, coniferyl alcohol, and sinapyl alcohol. The chemical structure of each lignin monomer includes a characteristic phenol group that contributes stability to the assumed radical intermediate formed upon oxidation. In this work, immobilized horseradish peroxidase was used to oxidize the coniferyl alcohol and to directly detect the radical intermediate using immobilized enzyme - ESR spectroscopy (IE-ESR). Oxidation products were analyzed by HPLC and findings will be discussed in terms of both lignin and lignan synthesis.
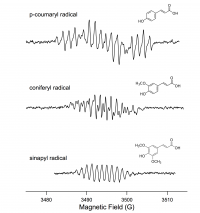
Preliminary ESR data
Lignin monomer radicals were detected using 50/50 water/dioxane (pH 5) to solublize polymer products; immobilized HRP (Figure 1). Quantum calculations to analyze each spectra are in progress; however, for now it can most definitely be concluded that each monomer has the ability to form radicals under oxidative conditions.
Oxidation of Coniferyl Alcohol
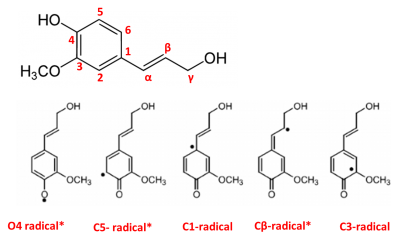
The chemical structure of coniferyl alcohol allows for various places where coupling may occur once the radical forms (Image A). Coniferyl alcohol was oxidized in a beaker under various reaction conditions to gain insight into the number of products formed by these radical-radical coupling reactions. A standard solution of the coniferyl alcohol monomer was made by dissolving 0.0385g of the product in 100 mL of a 50/50 dioxane/pH5 buffer. The first reaction was carried out with 5 mL of the stock solution (final concentration of 2mM coniferyl alcohol) in the presence of 10 microliters of HRP catalyst. The second reaction was carried out with 5 mL coniferyl alcohol in the presence of both 10 microliters HRP and 5 microliters of a 0.5M hydrogen peroxide solution (0.5 mM final concentration hydrogen peroxide). Subsequently, a third reaction was carried out involving 5 mL of coniferyl alcohol, 10 microliters of catalase, and 5 microliters of HRP. In the latter reaction, catalase was added last to the reaction beaker.
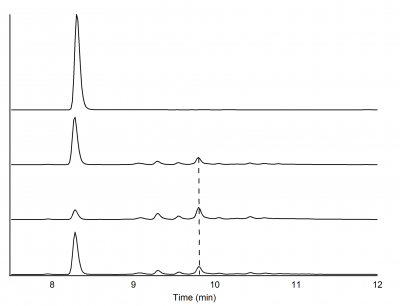
HPLC Analysis
Each reaction, along with the standard coniferyl alcohol solution, was analyzed using HPLC with an ACN/H20 gradient run for 35 minutes. The first 10 minutes were ran at 100% H20, followed by 80%ACN/20%H20 for minutes 10-30, and finishing from minutes 30-35 at 100% H20 (Figure 2). The exact method is saved in the HLPC instrument as "pcou_100417_35_SAZT."
Dicussion
This work is still in progress. Zelinda Taylor will be continuing the project as a Doc Kieft Scholar during the summer of 2018.
Poster
The following poster was presented by Stephanie Saey at the National ACS Meeting in New Orleans, Louisiana during the Spring of 2018:
References
Heitner, C., Dimmel, D.R., and Schmidt, J. (2010). Lignin and lignans: advances in chemistry.
Li X, Yang Z, Zhao M. (2012). Neuroprotective effects of Flax Lignan against NMDA‐induced neurotoxicity in vitro. CNS Neuroscience & Therapeutics, 18(11).927-933.
Vanholm, R., Demedts, B., Morreel, K., Ralph, J., and Boerjan, W. Lignin biosynthesis and structure. (2010). Plant Physiology. doi:10.1104/pp.110.155119