Difference between revisions of "Oxidation of Acetaminophen"
Jump to navigation
Jump to search
| Line 8: | Line 8: | ||
!Nuclei!!hyperfine (number spins) | !Nuclei!!hyperfine (number spins) | ||
|- | |- | ||
| − | | bgcolor=gray ! | + | | bgcolor=gray |
| + | ! !!Mason | ||
|- | |- | ||
| N || 0.81 G (1) | | N || 0.81 G (1) | ||
Revision as of 14:57, 15 July 2018
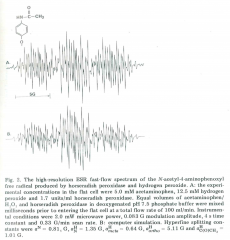
Current HRP/H2O2/APAP Radical Data
| Nuclei | hyperfine (number spins) | |
|---|---|---|
| bgcolor=gray | Mason | |
| N | 0.81 G (1) | |
| H, on nitrogen | 1.35 G (1) | |
| H, ortho | 5.11 G (1) | |
| H, meta | 0.64 G (1) | |
| H, methoxy | 1.01 (3) |
HRP/H2O2
- Free-Radical Metabolites of Acetaminophen and a Dimethylated Derivative
- Volker Fischer, Paul R. West, Laura S. Harman, and Ronald P. Mason.
- Environmental Health Perspectives, 1985, 64, pp. 127-137.
- ACETAMINOPHEN - ENZYMATIC FORMATION OF A TRANSIENT PHENOXYL FREE-RADICAL
- West, PR, Harman, LS, Josephy, PD, Mason, RP
- Biochemical pharmacology, 1984, Vol.33(18), pp.2933-2936.
- Direct electron spin resonance detection of free radical intermediates during the peroxidase catalyzed oxidation of phenacetin metabolites.
- Volker Fischer, Laura S. Harman, Paul R. West, and Ronald P. Mason.
- Chem.-Biol. Interactions, 1986, Vol 60, PP. 115-127.
HRP/H2O2/Acetaminophen analog
- Stable Free Radical and Benzoquinone Imine Metabolites of an Acetaminophen Analogue
- Volker Fischer and Ronald P. Mason.
- Journal of Biological Chemistry, 1984, 259(16), pp. 10284-10288.
- here
with GSH
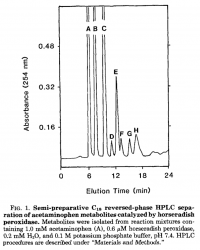
Oxidation Product Analysis
- Identification of Acetaminophen Polymerization Products Catalyzed by Horseradish Peroxidase.
- David W. Potter$, Dwight W. Miller, and Jack A. Hinson
- Journal of Biological Chemistry, 1985 "260(22), pp 12174-12180.
- here