Difference between revisions of "Lignin Monomers: Oxidation and Degradation"
| (161 intermediate revisions by 2 users not shown) | |||
| Line 1: | Line 1: | ||
| + | '''''The following document was written by Zelinda Taylor as partial fulfillment of her graduation requirements for a Bachelors Degree of Biochemistry, Monmouth College, spring 2020.''''' | ||
==Abstract== | ==Abstract== | ||
| − | The lignin polymer is synthesized via oxidative coupling of three basic monomers: p-coumaryl alcohol, coniferyl alcohol, and sinapyl alcohol. These three monomers each possess a phenol group that stabilizes the radical intermediate generated during the oxidation reaction. Although some literature exists showing the ESR spectra of these short-lived radical intermediates, we have revisited these intermediates using a combination of experimental techniques [HPLC and immobilized enzyme-ESR technique (IE-ESR)] and computational approaches [B3YLP/ESR-II and WINSIM]. | + | The lignin polymer is synthesized via oxidative coupling of three basic monomers: p-coumaryl alcohol, coniferyl alcohol, and sinapyl alcohol. These three monomers each possess a phenol group that stabilizes the radical intermediate generated during the oxidation reaction. Although some literature exists showing the ESR spectra of these short-lived radical intermediates, we have revisited these intermediates using a combination of experimental techniques [HPLC and immobilized enzyme-ESR technique (IE-ESR)] and computational approaches [B3YLP/ESR-II and WINSIM]. When two radicals dimerize together they initially create lignan, which is biologically active. HPLC was used to study the oxidation of coniferyl alcohol, the validate the formation of dimers. The mechanisms of this dimer formation were also studied to better understand the oxidative coupling of the lignin monomers. |
==Introduction== | ==Introduction== | ||
| − | + | ||
===Lignin=== | ===Lignin=== | ||
| + | |||
| + | |||
| + | Lignin is a large polymer structure in plant cell walls. The interest in lignin arose from the difficultly to isolate it from plants for products and structure determination<ref>[[:Media:Dimmel Overview.pdf|Dimmel, D. (2010). Overview. ''Lignin and Lignans Advances in Chemistry.'' Taylor & Francis Group.]]</ref>. This difficult process has produced a vast amount of research to determine how lignin is formed, and procedures to isolate it from plants without modifications. The issue lies with the lignin-carbohydrate linkages that occur between lignin and polymeric carbohydrates <ref>[[:Media:Dimmel Overview.pdf|Dimmel, D. (2010). Overview. ''Lignin and Lignans Advances in Chemistry.'' Taylor & Francis Group.]]</ref>. The difficulty associated with separating lignin from these carbohydrates and other matter of the plant makes trials on them difficult. Synthetic lignans are often used in research since the process of separating lignin is difficult and time consuming. The main purpose of lignin in plants is to assist water movement and distribution around the plant <ref>[[:Media:Dimmel Overview.pdf|Dimmel, D. (2010). Overview. ''Lignin and Lignans Advances in Chemistry.'' Taylor & Francis Group.]]</ref>. This property makes its presence especially important in trees. In softwood trees, it makes up about 28% of the mass of the tree <ref>[[:Media:Dimmel Overview.pdf|Dimmel, D. (2010). Overview. ''Lignin and Lignans Advances in Chemistry.'' Taylor & Francis Group.]]</ref>. It is the second most abundant polymer in the makeup of plants, cellulose being the first most abundant. There are three monomers of lignan: ferulic acid, p-coumaric acid, and sinapic acid. These are all characterized by a phenol group and a varying number of methoxy substituents occurring at the C-3 and C-5 positions. This phenol contributes to stability of the assumed radical forms due to resonance stabilization. There are alcohol forms of the monomers, which is what predominates in nature. These forms are coniferyl alcohol, p-coumaryl alcohol, and sinapyl alcohol. | ||
| + | |||
| + | <gallery class="center"> | ||
| + | File:ferulic acid poster.png|''Ferulic Acid'' | ||
| + | File:coniferyl alc poster.png|''Coniferyl Alcohol'' | ||
| + | File:sinapic poster.png|''Sinapic Acid'' | ||
| + | File:sinapyl alc poster.png|''Sinapyl Alcohol'' | ||
| + | File:p-coumaric acid.png|''p-coumaric Acid'' | ||
| + | File:P-coumaryl.jpg|''p-coumaryl Alcohol'' | ||
| + | </gallery> | ||
| + | |||
| + | They differ from the acid forms by having an alcohol group opposed to a carboxylic acid group on their side chain. The monomers couple together as radicals after they have undergone a one electron and one hydrogen oxidation process. Radicals occur at the O-4, C-1, C-3, C-5, and C-β positions <ref>[[:Media:Dimmel Overview.pdf|Dimmel, D. (2010). Overview. ''Lignin and Lignans Advances in Chemistry.'' Taylor & Francis Group.]]</ref>. By losing one electron and one hydrogen, a radical molecule is formed that is unstable. To gain stability, it undergoes oxidative coupling with another radical. This coupling can result in the formation of dimers, known as lignan, or polymers, known as lignin, but is more prevalent for a monomer to couple with a growing polymer <ref>[[:Media:Dimmel Overview.pdf|Dimmel, D. (2010). Overview. ''Lignin and Lignans Advances in Chemistry.'' Taylor & Francis Group.]]</ref>. The reactivity of the radicals can be determined using radical densities. This can be completed using ESR and computational chemistry programs, such as Web M.O. and WINSIM. Using these programs can help pinpoint which position has a higher radical density, which allows determination of which position a radical is more likely to form. Previous research determined these radicals were more likely at the C-5 and C-β positions <ref>[[:Media:Oxidative Coupling during Lignin Polymerization.pdf|Russel et al. (1996). Oxidative Coupling during Lignin Polymerization Is Determined by Unpaired Electron Delocalization within Parent Phenylpropanoid Radicals. ''Archives of Biochemistry and Biophysics, 332''(2), 357-366.]]</ref>. The delocalization of the unpaired electron significantly effects its reactivity and product formation. The comparison of this delocalization and substituent makeup can be used to observe influences on the radical coupling reactions <ref>[[:Media:Oxidative Coupling during Lignin Polymerization.pdf|Russel et al. (1996). Oxidative Coupling during Lignin Polymerization Is Determined by Unpaired Electron Delocalization within Parent Phenylpropanoid Radicals. ''Archives of Biochemistry and Biophysics, 332''(2), 357-366.]]</ref>. | ||
| + | |||
| + | |||
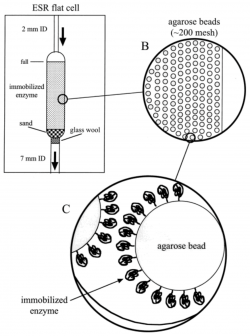
| + | ESR provides a basis for evaluating the radical formation of the monomers. Transient radicals, which are short lived and difficult to detect, are formed during the oxidation process <ref>[[:Media:Immobilized Enzyme ESR.pdf|Sturgeon, B. E., Chen, Y., & Mason, R. P. (2003). Immobilized Enzyme Electron Spin Resonance: A Method for Detecting Enzymatically Generated Transient Radicals. ''Analytical Chemistry, 75''(19), 5006-5011.]]</ref>. With the use of immobilized enzymes and ESR, these short lives radicals can be detected. The enzyme often used is horseradish peroxidase because of its low cost and availability. Other enzymes, such as lignin peroxidase, are able to function well in the oxidation reaction but possess more cost limitations <ref>[[:Media:Comparison of lignin peroxidase, horseradish peroxidase and laccase in the oxidation of methoxybenzenes.pdf|Kersten, P. J., Kalyanaraman, B., Hammel, K. E., Reinhammar, B., & Kirk, T. K. (1990). Comparison of lignin peroxidase, horseradish peroxidase and lactase in the oxidation of methoxybenzenes. ''Biochemical Journal, 268'', 475-480.]]</ref>. Lignin peroxidase is able to oxidize more methoxybenzenes, but HRP is effective as well due to its catalytic cycle being similar to lignin peroxidase <ref>[[:Media:Comparison of lignin peroxidase, horseradish peroxidase and laccase in the oxidation of methoxybenzenes.pdf|Kersten, P. J., Kalyanaraman, B., Hammel, K. E., Reinhammar, B., & Kirk, T. K. (1990). Comparison of lignin peroxidase, horseradish peroxidase and lactase in the oxidation of methoxybenzenes. ''Biochemical Journal, 268'', 475-480.]]</ref>. Previous methods of detection are not able to detect these radicals because they couple quickly with other radicals in solution. Flowing substrate solution over immobilized enzyme allows for detection of these radicals on a sub millisecond time scale <ref>[[:Media:Immobilized Enzyme ESR.pdf|Sturgeon, B. E., Chen, Y., & Mason, R. P. (2003). Immobilized Enzyme Electron Spin Resonance: A Method for Detecting Enzymatically Generated Transient Radicals. ''Analytical Chemistry, 75''(19), 5006-5011.]]</ref>. This method termed immobilized enzyme ESR, or IE-ESR, does not use a large amount of enzyme and the enzyme can be reused <ref>[[:Media:Immobilized Enzyme ESR.pdf|Sturgeon, B. E., Chen, Y., & Mason, R. P. (2003). Immobilized Enzyme Electron Spin Resonance: A Method for Detecting Enzymatically Generated Transient Radicals. ''Analytical Chemistry, 75''(19), 5006-5011.]]</ref>. For a continuous flow ESR experiment, about 20 mg of enzyme is needed compared to 0.75 mg of enzyme for IE-ESR <ref>[[:Media:Immobilized Enzyme ESR.pdf|Sturgeon, B. E., Chen, Y., & Mason, R. P. (2003). Immobilized Enzyme Electron Spin Resonance: A Method for Detecting Enzymatically Generated Transient Radicals. ''Analytical Chemistry, 75''(19), 5006-5011.]]</ref>. Since the substrate is flowing over the enzyme opposed to enzyme being needed throughout the substrate, the amount of enzyme needed to catalyze the reaction is significantly decreased. It also allows for different substrates to be ran over the immobilized enzyme as long as washes of the enzyme with buffer occurs between each new substrate. This method was developed in response to continuous flow ESR. This method is costly and wasteful compared to IE-ESR. The set up of this reaction is demonstrated to the right. | ||
| + | |||
| + | |||
| + | [[File:Screen Shot 2020-04-27 at 2.31.00 PM.png|250px|thumb|IE-ESR flat cell setup.]] | ||
| + | |||
| + | The use of the glass wool and sand prevents the immobilized enzyme from being washed out of the flat cell while ensuring constant substrate flow. The setup allows for consumption of less enzyme, substrate, and buffer making experiments less costly and wasteful. Although there are many benefits to using IE-ESR, the beads can become sticky leading to buildup of product on the beads. This buildup then interferes with the signal being collected. To help mitigate this issue, dioxane can be used in conjunction with buffer in the substrate to help wash the beads as the substrate flows over them. The use of immobilized enzymes in ESR compared to a continuous flow ESR experiment shows similar results <ref>[[:Media:Immobilized Enzyme ESR.pdf|Sturgeon, B. E., Chen, Y., & Mason, R. P. (2003). Immobilized Enzyme Electron Spin Resonance: A Method for Detecting Enzymatically Generated Transient Radicals. ''Analytical Chemistry, 75''(19), 5006-5011.]]</ref>. | ||
| + | |||
| + | |||
| + | The structure of lignin was determined after isolating lignin and degrading it into pieces <ref>[[:Media:Dimmel Overview.pdf|Dimmel, D. (2010). Overview. ''Lignin and Lignans Advances in Chemistry.'' Taylor & Francis Group.]]</ref>. By putting these pieces back together, the larger polymer structure was formulated. New methods, such as degradation, using thioacidolysis, are now being approached <ref>Lapierre, C. (2010). Determining Lignin Structure by Chemical Degradations. Lignin and Lignans Advances in Chemistry. Taylor & Francis Group.</ref>. There is an abundance of research that exists on lignin because of the challenges posed with isolating and studying it <ref>[[:Media:Dimmel Overview.pdf|Dimmel, D. (2010). Overview. ''Lignin and Lignans Advances in Chemistry.'' Taylor & Francis Group.]]</ref>. There are still many unknowns surrounding lignans due to its complex structure and varying coupling capabilities. | ||
===Lignan=== | ===Lignan=== | ||
| − | |||
| − | Research is currently being done to study the relationship between cancer and lignan. Breast cancer has been studied the most, but other cancers such as prostate and ovarian are being researched. Currently there are no conclusions regarding those cancers, as the research is in beginning stages. Many studies are centered around whether consumption of flax seeds correlates to a reduction in the risk of breast cancer, as well as the mortality. The reason flax seeds are being researched is because they have the highest concentration of lignans. With breast cancer, it was found flax seed consumption did decrease the risk of breast cancer, as well as the mortality of breast cancer, in post-menopausal women. This is believed to be because the lignan acts as a competitive inhibitor to estrogen. Some lignans, such as matairesinol and lariciresinol, have structures similar to estrogen. This allows them to bind to the estrogen receptor on the cancer cell, therefore blocking estrogen from binding. This prevents further growth of the cancer. | + | |
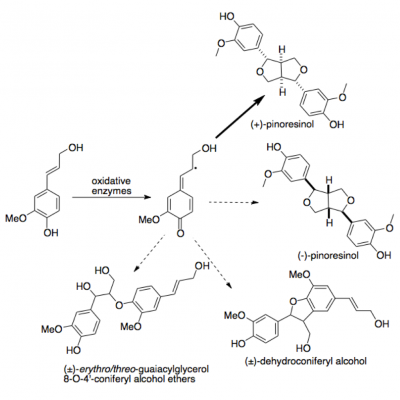
| + | The monomers of lignin can coupled together to form dimers, known as lignan. Lignan is formed from the coupling of two lignin monomers, often linked between the beta-carbons on the side chain of each monomer <ref>[[:Media:Lignan and Lignan Biosynthesis.pdf|Lewis, N. G., Davin, L. B., & Sarkanen, S. (1998) Lignan and Lignan Biosynthesis: Distinctions and Reconciliations. ''American Chemical Society Symposium Series'', 1-27.]]</ref>. They are used in plants as a means of defense and survival <ref>[[:Media:naturally lignan-rich foods.pdf|Rodríguez-García et al. (2019). Naturally Lignan-Rich Foods: A Dietary Tool for Health Promotion. ''Molecules, 24''(917), 1-25.]]</ref>. Many of the dimers expressed in literature possess this specific linking structure opposed to the more varying coupling of lignin. Other coupling can occur, but is less prominent in nature. These dimers have biological activity, whereas the larger structure of lignin does not. This allows them to have potential healths benefits associated with them. Some of these benefits are anti-microbial, anti-inflammatory, and antioxidant <ref>[[:Media:Flaxseed.pdf|Warwick, K. (2020). How Healthful is Flaxseed? ''Medical News Today.'']]</ref>. The relationship between allergies and lignan has also been studied <ref>[[:Media:Flaxseed.pdf|Warwick, K. (2020). How Healthful is Flaxseed? ''Medical News Today.'']]</ref>. It might be related to having an anti-histamine effect on allergy symptoms. Research is also being conducted on the activity of lignan with influenza <ref>[[:Media:Lignan and Pharmaceuticals.pdf|Kernan et al. (1997). Two New Lignans with Activity against Influenza Virus from the Medical Plant ''Rhinacanthus nasutus.'' ''Journal of Natural Products, 60''(6), 635-637.]]</ref>. Two new lignans have been isolated from ''Rhinacanthus nasutus'' and ''in vitro'' studies conducted. This plant is already used for treatments for illnesses such as fungal infections and herpes virus infections <ref>[[:Media:Lignan and Pharmaceuticals.pdf|Kernan et al. (1997). Two New Lignans with Activity against Influenza Virus from the Medical Plant ''Rhinacanthus nasutus.'' ''Journal of Natural Products, 60''(6), 635-637.]]</ref>. Lignans have the potential to become a contender in modern medicine for both prevention and treatment. | ||
| + | |||
| + | |||
| + | [[File:oxidative enzymes.png|400px|thumb|Beta-carbon linkages of different lignan compounds of coniferyl alcohol.]] | ||
| + | Research is currently being done to study the relationship between cancer and lignan <ref>[[:Media:naturally lignan-rich foods.pdf|Rodríguez-García et al. (2019). Naturally Lignan-Rich Foods: A Dietary Tool for Health Promotion. ''Molecules, 24''(917), 1-25.]]</ref>. Breast cancer has been studied the most, but other cancers such as prostate and ovarian are being researched. Currently there are no conclusions regarding those cancers, as the research is in beginning stages. Many studies are centered around whether consumption of flax seeds correlates to a reduction in the risk of breast cancer, as well as the mortality <ref>[[:Media:naturally lignan-rich foods.pdf|Rodríguez-García et al. (2019). Naturally Lignan-Rich Foods: A Dietary Tool for Health Promotion. ''Molecules, 24''(917), 1-25.]]</ref>. The reason flax seeds are being researched is because they have the highest concentration of lignans <ref>[[:Media:Flaxseed.pdf|Warwick, K. (2020). How Healthful is Flaxseed? ''Medical News Today.'']]</ref>. With breast cancer, it was found flax seed consumption did decrease the risk of breast cancer, as well as the mortality of breast cancer, in post-menopausal women <ref>[[:Media:naturally lignan-rich foods.pdf|Rodríguez-García et al. (2019). Naturally Lignan-Rich Foods: A Dietary Tool for Health Promotion. ''Molecules, 24''(917), 1-25.]]</ref>. This is believed to be because the lignan acts as a competitive inhibitor to estrogen. Some lignans, such as matairesinol and lariciresinol, have structures similar to estrogen. This allows them to bind to the estrogen receptor on the cancer cell, therefore blocking estrogen from binding <ref>[[:Media:naturally lignan-rich foods.pdf|Rodríguez-García et al. (2019). Naturally Lignan-Rich Foods: A Dietary Tool for Health Promotion. ''Molecules, 24''(917), 1-25.]]</ref>. This prevents further growth of the cancer. | ||
| + | |||
| + | |||
| + | Although lignans are formed via the coupling of two radical lignin monomers, other modifications are completed on the structure after coupling. Some of these are oxidations, reductions, and demethylations <ref>[[:Media:Lignan and Lignan Biosynthesis.pdf|Lewis, N. G., Davin, L. B., & Sarkanen, S. (1998) Lignan and Lignan Biosynthesis: Distinctions and Reconciliations. ''American Chemical Society Symposium Series'', 1-27.]]</ref>. Since there is strong similarity between lignans and lignin, it is difficult to separate their biochemical pathways <ref>[[:Media:Lignan and Lignan Biosynthesis.pdf|Lewis, N. G., Davin, L. B., & Sarkanen, S. (1998) Lignan and Lignan Biosynthesis: Distinctions and Reconciliations. ''American Chemical Society Symposium Series'', 1-27.]]</ref>. The distinction between the two has been studied greatly. It was determined the coupling process is dependent on plant species themselves. The coupling pathway differs between plant species due to proteins that are responsible for the mode of coupling in the formation of lignan <ref>[[:Media:Lignan and Lignan Biosynthesis.pdf|Lewis, N. G., Davin, L. B., & Sarkanen, S. (1998) Lignan and Lignan Biosynthesis: Distinctions and Reconciliations. ''American Chemical Society Symposium Series'', 1-27.]]</ref>. In the first plant species studied, ''Forsythia'', the protein was activated in the presence of an oxidase <ref>[[:Media:Lignan and Lignan Biosynthesis.pdf|Lewis, N. G., Davin, L. B., & Sarkanen, S. (1998) Lignan and Lignan Biosynthesis: Distinctions and Reconciliations. ''American Chemical Society Symposium Series'', 1-27.]]</ref>. Further research has shown the radical coupling and modifications are under explicit biochemical control <ref>[[:Media:Lignan and Lignan Biosynthesis.pdf|Lewis, N. G., Davin, L. B., & Sarkanen, S. (1998) Lignan and Lignan Biosynthesis: Distinctions and Reconciliations. ''American Chemical Society Symposium Series'', 1-27.]]</ref>. Lignans are more common in certain types of woods, as well as different cell types and tissues of plants. The same can be said for lignin, as its composition is higher in softwood trees compared to hardwood trees. The structures of these two molecules are similar, but their biochemical pathways are fully independent of one another <ref>[[:Media:naturally lignan-rich foods.pdf|Rodríguez-García et al. (2019). Naturally Lignan-Rich Foods: A Dietary Tool for Health Promotion. ''Molecules, 24''(917), 1-25.]]</ref>. This leaves the question of how do these lignans form and how do their pathways differ? | ||
| + | |||
| + | |||
| + | To determine the pathway of formation of these lignans, methods using enzymatic and chemical degradations have been approached. The use of enzymatic degradation would use enzymes to break down the larger structure into smaller, identifiable structures to determine how they piece together. Chemical degradations use reaction conditions to maximize the degradation of the larger structure to identify the parts. One of these is the use of thioacidolysis <ref>Lapierre, C. (2010). Determining Lignin Structure by Chemical Degradations. Lignin and Lignans Advances in Chemistry. Taylor & Francis Group.</ref>. It involves ether cleavage of the β-O-4 position <ref>Lapierre, C. (2010). Determining Lignin Structure by Chemical Degradations. Lignin and Lignans Advances in Chemistry. Taylor & Francis Group.</ref>. It involves EtSH and BF<sub>3</sub> etherate which allows for an increase in reaction yield <ref>Lapierre, C. (2010). Determining Lignin Structure by Chemical Degradations. Lignin and Lignans Advances in Chemistry. Taylor & Francis Group.</ref>. It was widely accepted due to its informative capabilities and simplicity <ref>Lapierre, C. (2010). Determining Lignin Structure by Chemical Degradations. Lignin and Lignans Advances in Chemistry. Taylor & Francis Group.</ref>. This process allows for quantification of the C<sub>6</sub>C<sub>3</sub> products. With the use of desulfurization, the lignans of the reaction can also be quantified. This could provide information on the coupling of the dimers compared to the larger structure of lignin. Other methods have involved placing lignin in a highly alkaline solution and to degrade and analyze the products. Methods exist for how to quantify lignans, but results are inconclusive. | ||
==Experimental Methods== | ==Experimental Methods== | ||
| Line 26: | Line 62: | ||
====Setup of Enzyme Immobilization==== | ====Setup of Enzyme Immobilization==== | ||
| − | + | A 0.190 mM immobilized enzyme solution (abs 1.94) was made using a simple apparatus. A Poly-Prep® Chromatography Column (Bio-Rad Laboratories, Inc. product 731-1550) and 3-way stopcock (Bio-Rad Laboratories, Inc. product 732-8103) were used. The column was secured to a ring stand and the 3-way stopper was attached. A 5 mL lure-lock syringe was attached to the 3-way stopcock at a 90° angle. The initial HRP solution is a 5 mM solution of peroxidase from horseradish and MOPS buffer. About 1 mL of Affi-Gel® 10 beads were added to the column attached to the ring stand and washed with water. They were then washed with MOPS buffer. The peroxidase and buffer solution in the eppendorf tube was added to the column containing the Affi-Gel® 10 beads and mixed by inverting and flicking the bottom of the column. The column was sealed and placed on a LabQuake® for an hour. | |
===Oxidation of Lignin Monomers Using Immobilized HRP=== | ===Oxidation of Lignin Monomers Using Immobilized HRP=== | ||
| Line 33: | Line 69: | ||
====Flat Cell Reactions with Immobilized Enzyme==== | ====Flat Cell Reactions with Immobilized Enzyme==== | ||
| − | A flat cell was loaded with glass wool, sand, and immobilized enzyme. Glass wool was lightly packed into the bottom neck of the flat cell. A small amount of sand was then mixed with nanopure water and added into the flat cell about 2 mm above the glass wool using a plastic 1 mL syringe. About 50 μL of immobilized enzyme is added on top of the glass wool and sand to fill most of the cavity of the flat cell. The pH 5 buffer was ran through the flat cell for about 10 minutes. A 1 mM ferulic acid, 1 mM hydrogen peroxide solution in 50/50 dioxane/pH 5 buffer was made. This solution was put in a 60 mL plastic syringe and flow controlled | + | A flat cell was loaded with glass wool, sand, and immobilized enzyme. Glass wool was lightly packed into the bottom neck of the flat cell. A small amount of sand was then mixed with nanopure water and added into the flat cell about 2 mm above the glass wool using a plastic 1 mL syringe. About 50 μL of immobilized enzyme is added on top of the glass wool and sand to fill most of the cavity of the flat cell. The pH 5 buffer was ran through the flat cell for about 10 minutes. A 1 mM ferulic acid, 1 mM hydrogen peroxide solution in 50/50 dioxane/pH 5 buffer was made. This solution was put in a 60 mL plastic syringe and flow was controlled using a syringe pump at 0.25 mL/minute, 0.5 mL/minute, 1 mL/minute, and 2 mL/minute. Flow was collected for each rate and analyzed on the HPLC. The same procedure was completed for p-coumaric acid. |
| − | === | + | ===ESR Analysis with Immobilized Enzyme=== |
| − | A flat cell was loaded with glass wool, sand, and immobilized enzyme using the procedure outlined above. A 10 mM ferulic acid solution in 50/50 dioxane/pH 7.4 buffer was made. Hydrogen peroxide was added to a concentration of 10 mM before | + | A flat cell was loaded with glass wool, sand, and immobilized enzyme using the procedure outlined above. A 10 mM ferulic acid solution in 50/50 dioxane/pH 7.4 buffer was made. Hydrogen peroxide was added to a concentration of 10 mM before ESR analysis. The solution was placed in a 60 mL plastic syringe and flow was controlled. Flow was analyzed at rates of 0.5 mL/minute, 1 mL/minute, and 2 mL/minute. The parameters for the program were modulation amplitude 0.5 G, sweep width 40.0 G, and center field at 3492.0 G. |
===Lignan Beaker Reactions=== | ===Lignan Beaker Reactions=== | ||
Beaker reactions of lignans were done with varying concentrations of hydrogen peroxide. Two lignan standards, (+)-pinoresinol and matiaresinol (purchased from Cayman Chemical, of 9.3 mM were created. A 2 mM coniferyl alcohol standard in 50/50 dioxane/pH 5 buffer was made. The first reaction was composed of 5 mL of substrate, 10 µL of 0.5 M hydrogen peroxide (1 mM final concentration hydrogen peroxide), and 5 µL of HRP. The second reaction was composed of 5 mL of substrate, 5 µL of 0.5 M hydrogen peroxide (0.5 mM final concentration hydrogen peroxide), and 5 µL of HRP. The third reaction was composed of 5 mL of substrate, 2.5 µL of 0.5 M hydrogen peroxide (0.25 mM final concentration hydrogen peroxide), and 5 µL of HRP. These reactions were then analyzed on the HPLC for lignan identification. | Beaker reactions of lignans were done with varying concentrations of hydrogen peroxide. Two lignan standards, (+)-pinoresinol and matiaresinol (purchased from Cayman Chemical, of 9.3 mM were created. A 2 mM coniferyl alcohol standard in 50/50 dioxane/pH 5 buffer was made. The first reaction was composed of 5 mL of substrate, 10 µL of 0.5 M hydrogen peroxide (1 mM final concentration hydrogen peroxide), and 5 µL of HRP. The second reaction was composed of 5 mL of substrate, 5 µL of 0.5 M hydrogen peroxide (0.5 mM final concentration hydrogen peroxide), and 5 µL of HRP. The third reaction was composed of 5 mL of substrate, 2.5 µL of 0.5 M hydrogen peroxide (0.25 mM final concentration hydrogen peroxide), and 5 µL of HRP. These reactions were then analyzed on the HPLC for lignan identification. | ||
| + | |||
| + | ====Lignin Analysis==== | ||
| + | The reactions and the standards were analyzed on the HPLC using an acetonitrile (ACN) and water gradient for 25 minutes. The first 10 minutes were ran at 100% water, 10-20 minutes were ran at an 80% ACN/20% water gradient, and the remaining 5 minutes at 100% water. | ||
===Lignin Degradation Using Alkaline Conditions=== | ===Lignin Degradation Using Alkaline Conditions=== | ||
| Line 66: | Line 105: | ||
==Results== | ==Results== | ||
| + | |||
| + | ===Lignin=== | ||
| + | ====HPLC Analysis==== | ||
| + | {| | ||
| + | [[File: 060118_data.jpg|275px|thumb|left|Figure 1: HPLC analysis of oxidation of ferulic acid with HRP and H<sub>2</sub>O<sub>2</sub> using 50/50 dioxane/pH 5 buffer.]] | ||
| + | || | ||
| + | [[File: 060718_data.jpg|275px|thumb|center|Figure 2: HPLC analysis of oxidation of p-coumaric acid with HRP and H<sub>2</sub>O<sub>2</sub> using 50/50 dioxane/pH 5 buffer.]] | ||
| + | || | ||
| + | [[File: 060818_data.jpg|275px|thumb|right|Figure 3: HPLC analysis of oxidation of synapic acid with HRP and H<sub>2</sub>O<sub>2</sub> using 50/50 dioxane/pH 5 buffer.]] | ||
| + | |} | ||
| + | |||
| + | |||
| + | In ''figure 1'', ''figure 2'', and ''figure 3'', beaker reactions of the oxidation of the three lignin monomers using H<sub>2</sub>O<sub>2</sub> was completed. The same procedure was done using a 2 mM solution of the standard in 50/50 dioxane and pH 5 buffer. Dioxane was introduced into the reaction to assist with possible product formation solubility. Each reaction consisted of 5 mL of substrate, 5 μL of HRP, and a varying amount of 0.5 M H<sub>2</sub>O<sub>2</sub> to reach final concentrations of 0.25 mM, 0.5 mM, and 1.0 mM. In ''figure 1'' and ''figure 2'', a standard is being consumed and producing oxidation products. These products could be dimers or small polymers. As the concentration of H<sub>2</sub>O<sub>2</sub> increases, the more the standard is consumed and the more products formed. Since each reaction only differed by the concentration of the H<sub>2</sub>O<sub>2</sub>, each reaction can be compared. In ''figure 3'' sinapic acid did not produce product peaks because the standard was never consumed. By changing the substrate to only being in pH 5 buffer, product peaks were seen. | ||
| + | |||
| + | |||
| + | {| | ||
| + | [[File: FA_HRPi.jpg|275px|thumb|left|Figure 4: HPLC analysis of oxidation of ferulic acid with HRP<sub>i</sub> and H<sub>2</sub>O<sub>2</sub> using 50/50 dioxane/pH 5 buffer.]] | ||
| + | || | ||
| + | [[File: pcou_HRPi.jpg|275px|thumb|center|Figure 5: HPLC analysis of oxidation of p-coumaric acid with HRP<sub>i</sub> and H<sub>2</sub>O<sub>2</sub> using 50/50 dioxane/pH 5 buffer.]] | ||
| + | || | ||
| + | [[File: sin_HRPi.jpg|275px|thumb|right|Figure 6: HPLC analysis of oxidation of synapic acid with HRP<sub>i</sub> and H<sub>2</sub>O<sub>2</sub> using pH 5 buffer.]] | ||
| + | |} | ||
| + | |||
| + | |||
| + | In ''figure 4'', ''figure 5'', and ''figure 6'', the same beaker reactions were completed as above using immobilized HRP in agarose beads. HPLC analysis was completed to ensure the enzyme was immobilized on the beads. Reactions were also completed to show reproducibility of previously collected HPLC data of the monomers. These reactions used 5 mL of substrate, 5 μL of HRP<sub>i</sub>, and a varying amount of 0.5 M H<sub>2</sub>O<sub>2</sub> to reach final concentrations of 0.25 mM, 0.5 mM, and 1.0 mM. These figures showed the standard being decreased and oxidation products being formed. As the concentration of the H<sub>2</sub>O<sub>2</sub> increased, more standard was consumed and more oxidation products were formed. ''Figure 6'' is showing sinapic acid producing product peaks, but no peaks were shown in ''figure 3'' with sinapic acid. This is because the substrate was changed to only be in pH 5 buffer opposed to 50/50 dioxane and pH 5 buffer. | ||
| + | |||
{| | {| | ||
| − | | | + | [[File: FA_FC_062118.jpg|275px|thumb|left|Figure 7: HPLC data of flowing 50/50 pH 5 buffer/dioxane with ferulic acid and H<sub>2</sub>O<sub>2</sub> through a flat cell loaded with HRP<sub>i</sub>.]] |
| + | || | ||
| + | [[File: pCou_FC_062218.jpg|275px|thumb|center|Figure 8: HPLC data of flowing 50/50 pH 5 buffer/dioxane with p-coumaric acid and H<sub>2</sub>O<sub>2</sub> through a flat cell loaded with HRP<sub>i</sub>.]] | ||
| + | |} | ||
| + | |||
| + | |||
| + | To evaluate the use of the immobilized enzyme in a flat cell, samples of the waste were collected to ensure the immobilized enzyme was functioning in the flat cell. To do this, a flat cell was loaded with steel wool, sand, and agarose beads with HRPi. MOPS Buffer was then ran over the beads in the flat cell to wash them and ensure they were not dry. A substrate of 1 mM ferulic acid and 1 mM hydrogen peroxide in 50/50 dioxane/pH 5 buffer was then ran over the immobilized enzyme in the flat cell. The flow of the substrate was controlled using a syringe pump at 0.25 mL/min, 0.5 mL/min, 1 mL/min, and 2 mL/min. Samples of waste were then collected at each flow rate and analyzed on the HPLC. The results of ferulic acid are shown in ''figure 7.'' The same procedure was used for a substrate of 1 mM p-coumaric acid and 1 mM hydrogen peroxide in 50/50 dioxane/pH 5 buffer and is shown in ''figure 8''. | ||
| + | |||
| + | ====ESR Analysis==== | ||
| + | |||
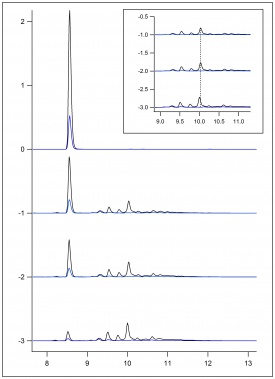
| + | [[File:Ligin Monomer radicals.png|300px|thumb|center|Figure 9: ESR data from HRP oxidation of lignin monomers]] | ||
| + | |||
| + | |||
| + | ''Figure 9'' shows previously collected ESR data of the oxidation of lignin monomers. In ''figure 10'', ''figure 11'', ''figure 12'' and ''figure 13'', preliminary ESR data is shown. To complete preliminary ESR data collection using ferulic acid, a substrate of 10 mM ferulic acid in 50/50 dioxane/pH 5 buffer was made. The flat cell was then loaded with steel wool, sand, and immobilized enzyme. Buffer was ran over the immobilized enzyme for about an hour. The flat cell was then mounted in the ESR. Hydrogen peroxide was added to the substrate directly before flow was began so its concentration in solution was 10 mM. The substrate was then flowed through the flat cell in the ESR at a flow rate of 1 mL/min and a signal was collected. The flow of the substrate was then increased to 2 mL/min and a signal was collected. The flow of the substrate was then stopped, and no signal was apparent. This was done to ensure the signal being produced was a result of the oxidation of the monomers. If this was true, stopping the flow would stop the signal. The flow was then re-started with a flow rate of 2 mL/min, and the signal returned. | ||
| + | |||
| + | |||
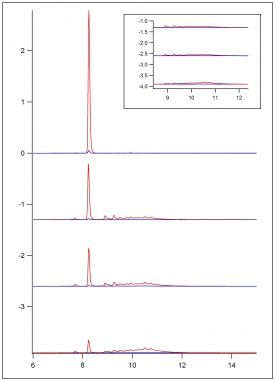
| + | {| | ||
| + | |[[File:FA_1_EPR_062518.jpg|275px|thumb|left|Figure 10: ESR data from substrate of ferulic acid and H<sub>2</sub>O<sub>2</sub> in 50/50 dioxane/pH 5 buffer with substrate flow of 1 mL/min.]] | ||
| + | || | ||
| + | [[File:FA_2_EPR_062518.jpg|275px|thumb|center|Figure 11: ESR data from substrate of ferulic acid and H<sub>2</sub>O<sub>2</sub> in 50/50 dioxane/pH 5 buffer with substrate flow of 2 mL/min.]] | ||
| + | || | ||
| + | [[File:FA_3_EPR_062518.jpg|275px|thumb|center|Figure 12: ESR data with no substrate flow.]] | ||
| + | || | ||
| + | [[File:FA_4_EPR_062518.jpg|275px|thumb|right|Figure 13: ESR data from substrate of ferulic acid and H<sub>2</sub>O<sub>2</sub> in 50/50 dioxane/pH 5 buffer with returning substrate flow of 2 mL/min.]] | ||
| + | |} | ||
| + | |||
| + | |||
| + | {| | ||
| + | |[[File:1_2_3.PNG|500px|thumb|left|Figure 14: ESR experimental collection & simulation from substrate of ferulic acid and H<sub>2</sub>O<sub>2</sub> in 50/50 dioxane/pH 7.4 buffer with substrate flow of 1 mL/min.]] | ||
| + | || | ||
| + | [[File:coniferyl alcohol_ESR.png|500px|thumb|right|Figure 15: ESR experimental collection & simulation from substrate of coniferyl alcohol and H<sub>2</sub>O<sub>2</sub> in 50/50 dioxane/pH 5 buffer with substrate flow of 1 mL/min.]] | ||
| + | |} | ||
| + | |||
| + | |||
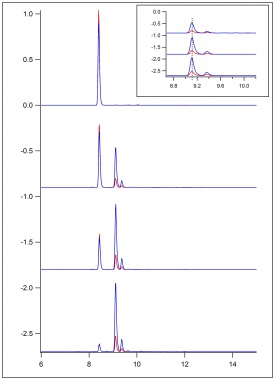
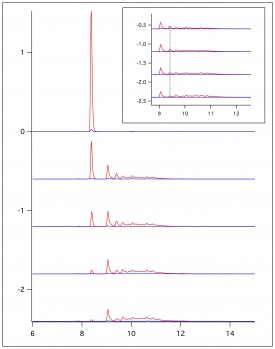
| + | Computational analyses were completed of ESR experimental data of ferulic acid and coniferyl alcohol. A computational program called WINSIM was used to compare experimental data with simulated data. Simulating the experimental data allowed for the determination of coupling constants of the assumed radicals formed. This allowed determination of which radicals were more likely to form and potentially couple together. | ||
| + | |||
| + | ====Comparison of Ferulic Acid and Coniferyl Alcohol Using WebMO Calculations & WINSIM Simulation==== | ||
| + | |||
| + | {| border=1 | ||
| + | |- bgcolor=grey | ||
| + | | ''Position'' || ''Coniferyl Alcohol'' (G03) ||''Coniferyl Alcohol'' (sim-Burrkitt) Chemical ox || ''Coniferyl Alcohol'' (sim) g-shift -0.26, LS 22, LW 0.32 || ''Ferulic Acid'' (G03) || ''Ferulic Acid'' (sim-Burkitt)|| ''Ferulic Acid'' (sim) g-shift: 1.46, LS: 80.9, LW: 0.34 | ||
| + | |- | ||
| + | |10 (ortho to -OH) || 5.01 || 5.33 ||5.52 || 4.99 || 5.56 ||4.09 | ||
|- | |- | ||
| + | |11 (meta to -OH) || 1.57 || 1.86 ||1.78 || 1.90 || 1.70 ||1.87 | ||
| + | |- | ||
| + | |20 (ortho to -OCH3) || 2.56 || 1.16 ||1.18 || 2.46 || 1.53 || 0.55 | ||
| + | |- | ||
| + | |19 (on alpha C || 2.49 || 2.32 || 3.08 || 2.71 || 2.64 || 2.62 | ||
| + | |- | ||
| + | |18 (on beta C) || 6.0 || 5.50 || 7.16|| 5.87 || 5.28 || 5.39 | ||
| + | |- | ||
| + | |17 (on gamma C|| 1.29 || 3.92 ||3.97 || x || x || x | ||
| + | |- | ||
| + | |21 (-OCH3) || 2.77 || 1.38 ||1.78 || 0.15 || 1.69 || 1.36 | ||
| + | |- | ||
| + | |22 (-OCH3)|| 0.15 || 1.38 || 1.78 || 2.54 || 1.69 || 1.36 | ||
| + | |- | ||
| + | |23 (-OCH3)|| 2.75 || 1.38 || 1.78 || 2.55 || 1.69 || 1.36 | ||
| + | |} | ||
| − | [[File: | + | |
| + | The above table shows the comparison of radical forms of coniferyl alcohol and ferulic acid. The table shows the experimental simulation values, previous collected values, and the WebMO 18 calculation values. These values collected at this institution are on the figures below, the experimental simulation values are in bold and the WebMO 18 calculation values are in standard text. | ||
| + | |||
| + | |||
| + | {| | ||
| + | |align=center|''Coniferyl Alcohol Radical''||align=center|''Ferulic Acid Radical''|| | ||
| + | |- | ||
| + | |||
| + | |||
| + | |[[File:Screen Shot 2019-04-22 at 10.13.27 AM.png|thumb|400px|Figure 16: Coniferyl alcohol radical with experimentally simulated and computed coupling constants.]] | ||
|| | || | ||
| + | [[File:Screen Shot 2019-04-22 at 10.13.22 AM.png|thumb|350px|Figure 17: Ferulic acid radical with experimentally simulated and computed coupling constants.]] | ||
| + | |} | ||
| − | [[File: | + | ===Lignan=== |
| + | |||
| + | ====HPLC Analysis==== | ||
| + | |||
| + | {| | ||
| + | |[[File:matiresinol.jpg|325px|thumb|left|Figure 18: Matiresinol in dioxane analysis using a water/acetonitrile gradient.]] | ||
| + | || | ||
| + | [[File:coniferyl alcohol.png|275px|thumb|center|Figure 19: Coniferyl alcohol analysis using varying concentrations of hydrogen peroxide using a water/acetonitrile gradient.]] | ||
|| | || | ||
| + | [[File:pinoresinol.jpg|325px|thumb|right|Figure 20: Pinoresinol in dioxane analysis using a water/acetonitrile gradient.]] | ||
| + | |} | ||
| − | + | ||
| − | + | Two dimers were used for analysis of the oxidation products of coniferyl alcohol. A 2mM standard of coniferyl alcohol in 50/50 dioxane and pH 5 buffer was used. A total of three reactions were done each with 5 mL of substrate, 5 μL of HRP, and a varying amount of 0.5 M H<sub>2</sub>O<sub>2</sub> to reach final concentrations of 0.25 mM, 0.5 mM, and 1.0 mM in the reaction. These reactions were analyzed on the HPLC and compared with the dimer standards of 9.3 mM. By using the dimer standards, it was hoped they could be identified in the oxidation products of the monomers since both dimers are dimers of coniferyl alcohol. Comparing the dimer standards to the oxidation products of the coniferyl alcohol, the dimers could not be identified. It is possible there are other dimers present in the oxidation products, but not these dimers. | |
==Discussion== | ==Discussion== | ||
| + | |||
| + | |||
| + | With the use of HRP and H<sub>2</sub>O<sub>2</sub>, lignan monomers were able to be oxidized and analyzed on the HPLC using an acetonitrile and water gradient. Concentration dependent reactions were made so the substrate decreased linearly as the concentration of H<sub>2</sub>O<sub>2</sub> within the reaction was increased. While the decrease of substrate was important, the products produced for each reaction were of interest. As the substrate was decreasing, or being consumed, the amount of products detected should increase. This was the case with the ferulic acid as well as the p-coumaric acid. For sinapic acid, this did not occur. The substrate was not consumed and products were not formed. Throughout each reaction, as the concentration of H<sub>2</sub>O<sub>2</sub> was increased, the same, prominent substrate peak was present. The lack of products could be due to the double-methoxy substitutions on the phenol. Ferulic acid only posses one, and p-coumaric acid possess none. It is possible the two methoxy groups of sinapic acid restrict oxidation, therefore forming no products. These same results were observed when using both HRP and immobilized HRP (HRP<sub>i</sub>) in beaker reactions. Once the substrate was limited to pH 5 buffer only, some product peaks formed opposed to none with the substrate in pH 5 buffer and dioxane. | ||
| + | |||
| + | |||
| + | Flat cell reactions were completed using HRP<sub>i</sub> and H<sub>2</sub>O<sub>2</sub> and analyzed on the HPLC to examine if oxidation products were being formed before analyzing the reactions using ESR. Products were produced meaning the HRP<sub>i</sub> was functioning and the reaction setup of the flat cell was adequate. Using the ESR for analysis, the coupling constants of the assumed radicals formed during the oxidation reaction were of interest. These constants could be determined using computational chemistry software, as well as simulations of the experimental data collected. Completing both of these analyses and comparing them showed radials were more likely to be formed at the C-5 and C-β positions than other positions on the molecule. These findings agreed with previous data collected by ''Russell, Forrester, Chesson, and Burkitt.'' The coupling constants of the assumed radials formed were of interest because they could be used as a way to predict how oxidative coupling occurs. The larger structure of lignin could be formed vs the dimers, known as lignan. By determining the coupling constants, the oxidative coupling process can be further studied to determine which structures are likely to couple together, and how they couple. | ||
| + | |||
| + | |||
| + | Lignan became of interest when analyzing oxidation products of the monomers on the HPLC. Their pathway of formation was of particular interest. The pathway was first approached using a process that started with the monomers and oxidized then. This was done because it was thought the radials formed during the oxidation reaction could potentially couple into dimers. Two lignans of coniferyl alcohol were chosen for analysis. By using these standards and comparing them to the beaker oxidation reactions of coniferyl alcohol in the presence of HRP and H<sub>2</sub>O<sub>2</sub>, these lignans could be identified in the oxidation products. This did not happen, but does not mean dimers did not exist in the products. The dimers chosen for analysis just were not present in the products. The pathway was then approached using a process that started with lignin, the larger structure, and attempted to break it down into dimers and monomers. This was done in an alkaline environment using NaOH. Results were inconclusive using this method, and stronger method development needs to be approached to determine the pathway of formation of the dimers from this angle. | ||
==Conclusion== | ==Conclusion== | ||
| + | |||
| + | |||
| + | Although some conclusions were made regarding the oxidation of lignin monomers, more research needs to be done to fully understand the oxidation process. Specifically, an analysis procedure needs to be determined for the products of the oxidation reaction. The oxidation reactions were successful, but no method was developed that allowed for successful analysis of the products of these reactions. Sinapic acid was also not oxidized, so a new approach needs to be done to oxidize this monomer and determine products of the reaction. | ||
| + | |||
| + | |||
| + | The EPR analysis of the monomers was successful as coupling constants were determined of the assumed radicals formed. This was only done with ferulic acid and coniferyl alcohol, so the other monomers need to be analyzed. It is likely they can be analyzed using the same method to determine these constants. It would be interesting to compare the acid and alcohol forms of the monomers for similarities in coupling constants. These constants can also then be compared among the monomers for similarities. | ||
| + | |||
| + | |||
| + | Little progress was made on the investigation into the pathway of formation of lignan. Two different approaches were used, but no conclusive results were developed for either of these. The degradation method presented the most promising results and literature is relatively abundant on the topic. More literature review should be done to determine a degradation method that can be completed and then analyzed on the HPLC. A successful method will assist with determining how lignan forms. This pathway can then be compared to the pathway of formation of lignin. Other lignans could also be researched to identify oxidation products of the monomers that could be dimers. | ||
==Future Directions== | ==Future Directions== | ||
| Line 89: | Line 250: | ||
==References== | ==References== | ||
| + | |||
| + | <references/> | ||
| + | |||
| + | ==Personal Reflection: Progress and Knowledge== | ||
| + | |||
| + | |||
| + | Before I first started doing research with Stephanie Saey as a sophomore, I was under the impression you mixed compounds together and hoped for the best for research. My eyes were opened when I realized much more goes into research. In fact, more time is spent outside of the lab planning and analyzing than actually being in the lab. I learned method development is an important aspect of research. You need to go into the lab with a plan, and an alternate plan sometimes. My research experience before coming to Monmouth was so juvenile. I did science fair presentations on such silly things that I thought were so revolutionary and life changing at the time. I look back on my research here and think, I really did make the most out of my experience. I accomplished more than I ever thought I would, and I enjoyed my research here. Yes at times it was a pain, and I put it on the back burner, but I always had a passion for my research. I guess you could say I'm a lignin '''superfan''' now! I think greater growth, both as a student and as a researcher, occurred through my research failures. At first, failures felt like a slap in the face. But as my experience progressed, I realized these failures can be turned into successes. Failures mean you have successfully figured out something about your research doesn't work. It can guide you or direct you to the next step in your research. These "failures" pushed me to work harder and crave to find the explanation for different things I was interested in. I think being able to see this project grow from the start really cultivated my interest and love for this project. I have this curious and stubborn drive within me that continues to push me toward my goals and aspirations. The mentality I have leaving this research is completely opposite the mentality I had beginning it! Although there were some bumps along the way, I wouldn't change one thing! I have growth throughout this research, for the better, and have hopefully set it up well for the next person to take over. My experience here has been filled with my failures, but not once did I give up and lose sight at the finish. | ||
| + | |||
| + | |||
| + | '''Success is the ability to go from one failure to another with no loss of enthusiasm – Winston Churchill''' | ||
Latest revision as of 13:38, 2 August 2020
The following document was written by Zelinda Taylor as partial fulfillment of her graduation requirements for a Bachelors Degree of Biochemistry, Monmouth College, spring 2020.
Abstract
The lignin polymer is synthesized via oxidative coupling of three basic monomers: p-coumaryl alcohol, coniferyl alcohol, and sinapyl alcohol. These three monomers each possess a phenol group that stabilizes the radical intermediate generated during the oxidation reaction. Although some literature exists showing the ESR spectra of these short-lived radical intermediates, we have revisited these intermediates using a combination of experimental techniques [HPLC and immobilized enzyme-ESR technique (IE-ESR)] and computational approaches [B3YLP/ESR-II and WINSIM]. When two radicals dimerize together they initially create lignan, which is biologically active. HPLC was used to study the oxidation of coniferyl alcohol, the validate the formation of dimers. The mechanisms of this dimer formation were also studied to better understand the oxidative coupling of the lignin monomers.
Introduction
Lignin
Lignin is a large polymer structure in plant cell walls. The interest in lignin arose from the difficultly to isolate it from plants for products and structure determination[1]. This difficult process has produced a vast amount of research to determine how lignin is formed, and procedures to isolate it from plants without modifications. The issue lies with the lignin-carbohydrate linkages that occur between lignin and polymeric carbohydrates [2]. The difficulty associated with separating lignin from these carbohydrates and other matter of the plant makes trials on them difficult. Synthetic lignans are often used in research since the process of separating lignin is difficult and time consuming. The main purpose of lignin in plants is to assist water movement and distribution around the plant [3]. This property makes its presence especially important in trees. In softwood trees, it makes up about 28% of the mass of the tree [4]. It is the second most abundant polymer in the makeup of plants, cellulose being the first most abundant. There are three monomers of lignan: ferulic acid, p-coumaric acid, and sinapic acid. These are all characterized by a phenol group and a varying number of methoxy substituents occurring at the C-3 and C-5 positions. This phenol contributes to stability of the assumed radical forms due to resonance stabilization. There are alcohol forms of the monomers, which is what predominates in nature. These forms are coniferyl alcohol, p-coumaryl alcohol, and sinapyl alcohol.
They differ from the acid forms by having an alcohol group opposed to a carboxylic acid group on their side chain. The monomers couple together as radicals after they have undergone a one electron and one hydrogen oxidation process. Radicals occur at the O-4, C-1, C-3, C-5, and C-β positions [5]. By losing one electron and one hydrogen, a radical molecule is formed that is unstable. To gain stability, it undergoes oxidative coupling with another radical. This coupling can result in the formation of dimers, known as lignan, or polymers, known as lignin, but is more prevalent for a monomer to couple with a growing polymer [6]. The reactivity of the radicals can be determined using radical densities. This can be completed using ESR and computational chemistry programs, such as Web M.O. and WINSIM. Using these programs can help pinpoint which position has a higher radical density, which allows determination of which position a radical is more likely to form. Previous research determined these radicals were more likely at the C-5 and C-β positions [7]. The delocalization of the unpaired electron significantly effects its reactivity and product formation. The comparison of this delocalization and substituent makeup can be used to observe influences on the radical coupling reactions [8].
ESR provides a basis for evaluating the radical formation of the monomers. Transient radicals, which are short lived and difficult to detect, are formed during the oxidation process [9]. With the use of immobilized enzymes and ESR, these short lives radicals can be detected. The enzyme often used is horseradish peroxidase because of its low cost and availability. Other enzymes, such as lignin peroxidase, are able to function well in the oxidation reaction but possess more cost limitations [10]. Lignin peroxidase is able to oxidize more methoxybenzenes, but HRP is effective as well due to its catalytic cycle being similar to lignin peroxidase [11]. Previous methods of detection are not able to detect these radicals because they couple quickly with other radicals in solution. Flowing substrate solution over immobilized enzyme allows for detection of these radicals on a sub millisecond time scale [12]. This method termed immobilized enzyme ESR, or IE-ESR, does not use a large amount of enzyme and the enzyme can be reused [13]. For a continuous flow ESR experiment, about 20 mg of enzyme is needed compared to 0.75 mg of enzyme for IE-ESR [14]. Since the substrate is flowing over the enzyme opposed to enzyme being needed throughout the substrate, the amount of enzyme needed to catalyze the reaction is significantly decreased. It also allows for different substrates to be ran over the immobilized enzyme as long as washes of the enzyme with buffer occurs between each new substrate. This method was developed in response to continuous flow ESR. This method is costly and wasteful compared to IE-ESR. The set up of this reaction is demonstrated to the right.
The use of the glass wool and sand prevents the immobilized enzyme from being washed out of the flat cell while ensuring constant substrate flow. The setup allows for consumption of less enzyme, substrate, and buffer making experiments less costly and wasteful. Although there are many benefits to using IE-ESR, the beads can become sticky leading to buildup of product on the beads. This buildup then interferes with the signal being collected. To help mitigate this issue, dioxane can be used in conjunction with buffer in the substrate to help wash the beads as the substrate flows over them. The use of immobilized enzymes in ESR compared to a continuous flow ESR experiment shows similar results [15].
The structure of lignin was determined after isolating lignin and degrading it into pieces [16]. By putting these pieces back together, the larger polymer structure was formulated. New methods, such as degradation, using thioacidolysis, are now being approached [17]. There is an abundance of research that exists on lignin because of the challenges posed with isolating and studying it [18]. There are still many unknowns surrounding lignans due to its complex structure and varying coupling capabilities.
Lignan
The monomers of lignin can coupled together to form dimers, known as lignan. Lignan is formed from the coupling of two lignin monomers, often linked between the beta-carbons on the side chain of each monomer [19]. They are used in plants as a means of defense and survival [20]. Many of the dimers expressed in literature possess this specific linking structure opposed to the more varying coupling of lignin. Other coupling can occur, but is less prominent in nature. These dimers have biological activity, whereas the larger structure of lignin does not. This allows them to have potential healths benefits associated with them. Some of these benefits are anti-microbial, anti-inflammatory, and antioxidant [21]. The relationship between allergies and lignan has also been studied [22]. It might be related to having an anti-histamine effect on allergy symptoms. Research is also being conducted on the activity of lignan with influenza [23]. Two new lignans have been isolated from Rhinacanthus nasutus and in vitro studies conducted. This plant is already used for treatments for illnesses such as fungal infections and herpes virus infections [24]. Lignans have the potential to become a contender in modern medicine for both prevention and treatment.
Research is currently being done to study the relationship between cancer and lignan [25]. Breast cancer has been studied the most, but other cancers such as prostate and ovarian are being researched. Currently there are no conclusions regarding those cancers, as the research is in beginning stages. Many studies are centered around whether consumption of flax seeds correlates to a reduction in the risk of breast cancer, as well as the mortality [26]. The reason flax seeds are being researched is because they have the highest concentration of lignans [27]. With breast cancer, it was found flax seed consumption did decrease the risk of breast cancer, as well as the mortality of breast cancer, in post-menopausal women [28]. This is believed to be because the lignan acts as a competitive inhibitor to estrogen. Some lignans, such as matairesinol and lariciresinol, have structures similar to estrogen. This allows them to bind to the estrogen receptor on the cancer cell, therefore blocking estrogen from binding [29]. This prevents further growth of the cancer.
Although lignans are formed via the coupling of two radical lignin monomers, other modifications are completed on the structure after coupling. Some of these are oxidations, reductions, and demethylations [30]. Since there is strong similarity between lignans and lignin, it is difficult to separate their biochemical pathways [31]. The distinction between the two has been studied greatly. It was determined the coupling process is dependent on plant species themselves. The coupling pathway differs between plant species due to proteins that are responsible for the mode of coupling in the formation of lignan [32]. In the first plant species studied, Forsythia, the protein was activated in the presence of an oxidase [33]. Further research has shown the radical coupling and modifications are under explicit biochemical control [34]. Lignans are more common in certain types of woods, as well as different cell types and tissues of plants. The same can be said for lignin, as its composition is higher in softwood trees compared to hardwood trees. The structures of these two molecules are similar, but their biochemical pathways are fully independent of one another [35]. This leaves the question of how do these lignans form and how do their pathways differ?
To determine the pathway of formation of these lignans, methods using enzymatic and chemical degradations have been approached. The use of enzymatic degradation would use enzymes to break down the larger structure into smaller, identifiable structures to determine how they piece together. Chemical degradations use reaction conditions to maximize the degradation of the larger structure to identify the parts. One of these is the use of thioacidolysis [36]. It involves ether cleavage of the β-O-4 position [37]. It involves EtSH and BF3 etherate which allows for an increase in reaction yield [38]. It was widely accepted due to its informative capabilities and simplicity [39]. This process allows for quantification of the C6C3 products. With the use of desulfurization, the lignans of the reaction can also be quantified. This could provide information on the coupling of the dimers compared to the larger structure of lignin. Other methods have involved placing lignin in a highly alkaline solution and to degrade and analyze the products. Methods exist for how to quantify lignans, but results are inconclusive.
Experimental Methods
Oxidation of Lignin Monomers
Beaker reactions with varying reaction conditions were completed to analyze the oxidation of each monomer. For each monomer, 100 mL of a 2 mM standard solution of the monomer (purchased from Sigma Aldrich) was made using 50/50 dioxane/pH 5 buffer. The pH 5 buffer was a phosphate-citrate buffer (purchased from Sigma Aldrich). Three reactions were then completed with varying concentrations of hydrogen peroxide in the presence of a constant HRP concentration in scintillation vials. The first reaction was composed of 5 mL of the 2 mM substrate, 10 μL of 0.5 M hydrogen peroxide (1 mM final concentration hydrogen peroxide) and 5 μL of HRP. The second reaction was composed of 5 mL of the 2 mM substrate, 5 μL of 0.5 M hydrogen peroxide (0.5 mM final concentration hydrogen peroxide) and 5 μL of HRP. The third reaction was composed of 5 mL of the 2 mM substrate, 5 μL of 0.5 M hydrogen peroxide (0.25 mM final concentration hydrogen peroxide) and 5 μL of HRP. These reactions were then analyzed on the HPLC for oxidation products of each monomer and compared.
Analysis of Monomer Oxidation
The reactions and the standards were analyzed on the HPLC using an acetonitrile (ACN) and water gradient for 25 minutes. The first 10 minutes were ran at 100% water, 10-20 minutes were ran at an 80% ACN/20% water gradient, and the remaining 5 minutes at 100% water.
Immobilization of Enzymes
An immobilization of peroxidase from horseradish (HRP) was completed using Affi-Gel® 10. This procedure is centered around the isolectric point of the enzyme. The immobilization was completed using a MOPS buffer (pH 7.0) and about 1 mL of Affi-Gel® 10 beads.
Setup of Enzyme Immobilization
A 0.190 mM immobilized enzyme solution (abs 1.94) was made using a simple apparatus. A Poly-Prep® Chromatography Column (Bio-Rad Laboratories, Inc. product 731-1550) and 3-way stopcock (Bio-Rad Laboratories, Inc. product 732-8103) were used. The column was secured to a ring stand and the 3-way stopper was attached. A 5 mL lure-lock syringe was attached to the 3-way stopcock at a 90° angle. The initial HRP solution is a 5 mM solution of peroxidase from horseradish and MOPS buffer. About 1 mL of Affi-Gel® 10 beads were added to the column attached to the ring stand and washed with water. They were then washed with MOPS buffer. The peroxidase and buffer solution in the eppendorf tube was added to the column containing the Affi-Gel® 10 beads and mixed by inverting and flicking the bottom of the column. The column was sealed and placed on a LabQuake® for an hour.
Oxidation of Lignin Monomers Using Immobilized HRP
Beaker reactions with immobilized HRP
Beaker reactions were completed using the immobilized HRP and analyzed on the HPLC. The reactions had varying concentrations of hydrogen peroxide. A 2 mM standard was made for each monomer with a volume of 100 using 50/50 dioxane/pH 5 buffer. Before the immobilized HRP was used, it was rinsed twice with MOPS buffer. The first reaction was composed of 5 mL of substrate, 10 µL of 0.5 M hydrogen peroxide (1 mM final concentration hydrogen peroxide), and 50 µL of immobilized HRP. The second reaction was composed of 5 mL of substrate, 5 µL of 0.5 M hydrogen peroxide (0.5 mM final concentration hydrogen peroxide), and 50 µL of immobilized HRP. The third reaction was composed of 5 mL of substrate, 2.5 µL of 0.5 M hydrogen peroxide (0.25 mM final concentration hydrogen peroxide), and 50 µL of immobilized HRP. These reactions were then analyzed on the HPLC for possible oxidation products. The results were then compared to previously collected HPLC data using HRP, opposed to immobilized HRP.
Flat Cell Reactions with Immobilized Enzyme
A flat cell was loaded with glass wool, sand, and immobilized enzyme. Glass wool was lightly packed into the bottom neck of the flat cell. A small amount of sand was then mixed with nanopure water and added into the flat cell about 2 mm above the glass wool using a plastic 1 mL syringe. About 50 μL of immobilized enzyme is added on top of the glass wool and sand to fill most of the cavity of the flat cell. The pH 5 buffer was ran through the flat cell for about 10 minutes. A 1 mM ferulic acid, 1 mM hydrogen peroxide solution in 50/50 dioxane/pH 5 buffer was made. This solution was put in a 60 mL plastic syringe and flow was controlled using a syringe pump at 0.25 mL/minute, 0.5 mL/minute, 1 mL/minute, and 2 mL/minute. Flow was collected for each rate and analyzed on the HPLC. The same procedure was completed for p-coumaric acid.
ESR Analysis with Immobilized Enzyme
A flat cell was loaded with glass wool, sand, and immobilized enzyme using the procedure outlined above. A 10 mM ferulic acid solution in 50/50 dioxane/pH 7.4 buffer was made. Hydrogen peroxide was added to a concentration of 10 mM before ESR analysis. The solution was placed in a 60 mL plastic syringe and flow was controlled. Flow was analyzed at rates of 0.5 mL/minute, 1 mL/minute, and 2 mL/minute. The parameters for the program were modulation amplitude 0.5 G, sweep width 40.0 G, and center field at 3492.0 G.
Lignan Beaker Reactions
Beaker reactions of lignans were done with varying concentrations of hydrogen peroxide. Two lignan standards, (+)-pinoresinol and matiaresinol (purchased from Cayman Chemical, of 9.3 mM were created. A 2 mM coniferyl alcohol standard in 50/50 dioxane/pH 5 buffer was made. The first reaction was composed of 5 mL of substrate, 10 µL of 0.5 M hydrogen peroxide (1 mM final concentration hydrogen peroxide), and 5 µL of HRP. The second reaction was composed of 5 mL of substrate, 5 µL of 0.5 M hydrogen peroxide (0.5 mM final concentration hydrogen peroxide), and 5 µL of HRP. The third reaction was composed of 5 mL of substrate, 2.5 µL of 0.5 M hydrogen peroxide (0.25 mM final concentration hydrogen peroxide), and 5 µL of HRP. These reactions were then analyzed on the HPLC for lignan identification.
Lignin Analysis
The reactions and the standards were analyzed on the HPLC using an acetonitrile (ACN) and water gradient for 25 minutes. The first 10 minutes were ran at 100% water, 10-20 minutes were ran at an 80% ACN/20% water gradient, and the remaining 5 minutes at 100% water.
Lignin Degradation Using Alkaline Conditions
Lignin (purchased from Sigma Aldrich) was degraded in an alkaline solution of 100 mM NaOH. A solution of 50 grams of lignin and 10 mL of 100 mM NaOH were stirred on a stir plate for 24 hours. Samples of 10 µL of the mixture were extracted at 1 minute, 24 hours, and 48 hours after stirring and placed in 3 mL of pH 5 buffer. The samples were analyzed on the HPLC for degradation products.
Notes of Safety
Dioxane
Molecular Formula: C4H8O2
Molecular weight: 88.11 g/mol
Physical appearance: clear, colorless liquid
1,4-dioxane has the potential to be an eye, skin, and digestive tract irritant. To avoid contact, lab goggles, gloves, and protective clothing should be worn. In the case of exposure, eyes and skin should be flushed with water. If ingested, vomiting should not be induced and medical aid should be sought. Keep away from heat, sparks, and flame and use in a fume hood. Inhalation could induce breathing difficulties. It should be stored under nitrogen to less peroxide formation. If it is noticed that peroxide formation has occurred, do not open the container unless a professional. It has the potential to form explosive peroxides with prolonged exposure.
Sodium hydroxide
Molecular formula: NaOH
Molecular weight: 40 g/mol
Physical appearance: solid, white
Sodium hydroxide can cause eye, skin, and digestive tract burns. In the case of contact with sodium hydroxide, flush the eyes and skin with water for at least 15 minutes and seek medical attention. Vomiting should not be induced, but water can be drank. To mitigate these risks, lab safety googles, gloves, and protective clothing should be worn. Sodium hydroxide is hygroscopic so it must be stored in a tightly closed container. It is corrosive to material so caution is needed in case of a spill. Material should be disposed of in the proper disposal container.
Results
Lignin
HPLC Analysis
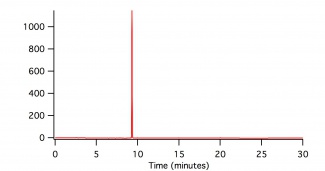
In figure 1, figure 2, and figure 3, beaker reactions of the oxidation of the three lignin monomers using H2O2 was completed. The same procedure was done using a 2 mM solution of the standard in 50/50 dioxane and pH 5 buffer. Dioxane was introduced into the reaction to assist with possible product formation solubility. Each reaction consisted of 5 mL of substrate, 5 μL of HRP, and a varying amount of 0.5 M H2O2 to reach final concentrations of 0.25 mM, 0.5 mM, and 1.0 mM. In figure 1 and figure 2, a standard is being consumed and producing oxidation products. These products could be dimers or small polymers. As the concentration of H2O2 increases, the more the standard is consumed and the more products formed. Since each reaction only differed by the concentration of the H2O2, each reaction can be compared. In figure 3 sinapic acid did not produce product peaks because the standard was never consumed. By changing the substrate to only being in pH 5 buffer, product peaks were seen.
In figure 4, figure 5, and figure 6, the same beaker reactions were completed as above using immobilized HRP in agarose beads. HPLC analysis was completed to ensure the enzyme was immobilized on the beads. Reactions were also completed to show reproducibility of previously collected HPLC data of the monomers. These reactions used 5 mL of substrate, 5 μL of HRPi, and a varying amount of 0.5 M H2O2 to reach final concentrations of 0.25 mM, 0.5 mM, and 1.0 mM. These figures showed the standard being decreased and oxidation products being formed. As the concentration of the H2O2 increased, more standard was consumed and more oxidation products were formed. Figure 6 is showing sinapic acid producing product peaks, but no peaks were shown in figure 3 with sinapic acid. This is because the substrate was changed to only be in pH 5 buffer opposed to 50/50 dioxane and pH 5 buffer.
To evaluate the use of the immobilized enzyme in a flat cell, samples of the waste were collected to ensure the immobilized enzyme was functioning in the flat cell. To do this, a flat cell was loaded with steel wool, sand, and agarose beads with HRPi. MOPS Buffer was then ran over the beads in the flat cell to wash them and ensure they were not dry. A substrate of 1 mM ferulic acid and 1 mM hydrogen peroxide in 50/50 dioxane/pH 5 buffer was then ran over the immobilized enzyme in the flat cell. The flow of the substrate was controlled using a syringe pump at 0.25 mL/min, 0.5 mL/min, 1 mL/min, and 2 mL/min. Samples of waste were then collected at each flow rate and analyzed on the HPLC. The results of ferulic acid are shown in figure 7. The same procedure was used for a substrate of 1 mM p-coumaric acid and 1 mM hydrogen peroxide in 50/50 dioxane/pH 5 buffer and is shown in figure 8.
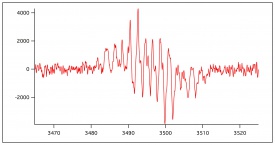
ESR Analysis
Figure 9 shows previously collected ESR data of the oxidation of lignin monomers. In figure 10, figure 11, figure 12 and figure 13, preliminary ESR data is shown. To complete preliminary ESR data collection using ferulic acid, a substrate of 10 mM ferulic acid in 50/50 dioxane/pH 5 buffer was made. The flat cell was then loaded with steel wool, sand, and immobilized enzyme. Buffer was ran over the immobilized enzyme for about an hour. The flat cell was then mounted in the ESR. Hydrogen peroxide was added to the substrate directly before flow was began so its concentration in solution was 10 mM. The substrate was then flowed through the flat cell in the ESR at a flow rate of 1 mL/min and a signal was collected. The flow of the substrate was then increased to 2 mL/min and a signal was collected. The flow of the substrate was then stopped, and no signal was apparent. This was done to ensure the signal being produced was a result of the oxidation of the monomers. If this was true, stopping the flow would stop the signal. The flow was then re-started with a flow rate of 2 mL/min, and the signal returned.
Computational analyses were completed of ESR experimental data of ferulic acid and coniferyl alcohol. A computational program called WINSIM was used to compare experimental data with simulated data. Simulating the experimental data allowed for the determination of coupling constants of the assumed radicals formed. This allowed determination of which radicals were more likely to form and potentially couple together.
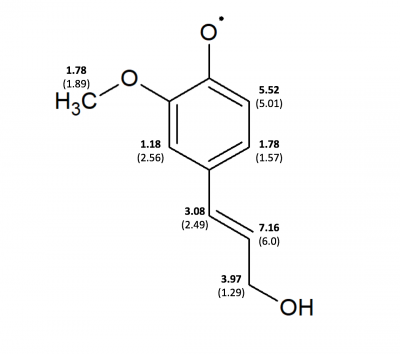
Comparison of Ferulic Acid and Coniferyl Alcohol Using WebMO Calculations & WINSIM Simulation
| Position | Coniferyl Alcohol (G03) | Coniferyl Alcohol (sim-Burrkitt) Chemical ox | Coniferyl Alcohol (sim) g-shift -0.26, LS 22, LW 0.32 | Ferulic Acid (G03) | Ferulic Acid (sim-Burkitt) | Ferulic Acid (sim) g-shift: 1.46, LS: 80.9, LW: 0.34 |
| 10 (ortho to -OH) | 5.01 | 5.33 | 5.52 | 4.99 | 5.56 | 4.09 |
| 11 (meta to -OH) | 1.57 | 1.86 | 1.78 | 1.90 | 1.70 | 1.87 |
| 20 (ortho to -OCH3) | 2.56 | 1.16 | 1.18 | 2.46 | 1.53 | 0.55 |
| 19 (on alpha C | 2.49 | 2.32 | 3.08 | 2.71 | 2.64 | 2.62 |
| 18 (on beta C) | 6.0 | 5.50 | 7.16 | 5.87 | 5.28 | 5.39 |
| 17 (on gamma C | 1.29 | 3.92 | 3.97 | x | x | x |
| 21 (-OCH3) | 2.77 | 1.38 | 1.78 | 0.15 | 1.69 | 1.36 |
| 22 (-OCH3) | 0.15 | 1.38 | 1.78 | 2.54 | 1.69 | 1.36 |
| 23 (-OCH3) | 2.75 | 1.38 | 1.78 | 2.55 | 1.69 | 1.36 |
The above table shows the comparison of radical forms of coniferyl alcohol and ferulic acid. The table shows the experimental simulation values, previous collected values, and the WebMO 18 calculation values. These values collected at this institution are on the figures below, the experimental simulation values are in bold and the WebMO 18 calculation values are in standard text.
| Coniferyl Alcohol Radical | Ferulic Acid Radical | |
Lignan
HPLC Analysis
Two dimers were used for analysis of the oxidation products of coniferyl alcohol. A 2mM standard of coniferyl alcohol in 50/50 dioxane and pH 5 buffer was used. A total of three reactions were done each with 5 mL of substrate, 5 μL of HRP, and a varying amount of 0.5 M H2O2 to reach final concentrations of 0.25 mM, 0.5 mM, and 1.0 mM in the reaction. These reactions were analyzed on the HPLC and compared with the dimer standards of 9.3 mM. By using the dimer standards, it was hoped they could be identified in the oxidation products of the monomers since both dimers are dimers of coniferyl alcohol. Comparing the dimer standards to the oxidation products of the coniferyl alcohol, the dimers could not be identified. It is possible there are other dimers present in the oxidation products, but not these dimers.
Discussion
With the use of HRP and H2O2, lignan monomers were able to be oxidized and analyzed on the HPLC using an acetonitrile and water gradient. Concentration dependent reactions were made so the substrate decreased linearly as the concentration of H2O2 within the reaction was increased. While the decrease of substrate was important, the products produced for each reaction were of interest. As the substrate was decreasing, or being consumed, the amount of products detected should increase. This was the case with the ferulic acid as well as the p-coumaric acid. For sinapic acid, this did not occur. The substrate was not consumed and products were not formed. Throughout each reaction, as the concentration of H2O2 was increased, the same, prominent substrate peak was present. The lack of products could be due to the double-methoxy substitutions on the phenol. Ferulic acid only posses one, and p-coumaric acid possess none. It is possible the two methoxy groups of sinapic acid restrict oxidation, therefore forming no products. These same results were observed when using both HRP and immobilized HRP (HRPi) in beaker reactions. Once the substrate was limited to pH 5 buffer only, some product peaks formed opposed to none with the substrate in pH 5 buffer and dioxane.
Flat cell reactions were completed using HRPi and H2O2 and analyzed on the HPLC to examine if oxidation products were being formed before analyzing the reactions using ESR. Products were produced meaning the HRPi was functioning and the reaction setup of the flat cell was adequate. Using the ESR for analysis, the coupling constants of the assumed radicals formed during the oxidation reaction were of interest. These constants could be determined using computational chemistry software, as well as simulations of the experimental data collected. Completing both of these analyses and comparing them showed radials were more likely to be formed at the C-5 and C-β positions than other positions on the molecule. These findings agreed with previous data collected by Russell, Forrester, Chesson, and Burkitt. The coupling constants of the assumed radials formed were of interest because they could be used as a way to predict how oxidative coupling occurs. The larger structure of lignin could be formed vs the dimers, known as lignan. By determining the coupling constants, the oxidative coupling process can be further studied to determine which structures are likely to couple together, and how they couple.
Lignan became of interest when analyzing oxidation products of the monomers on the HPLC. Their pathway of formation was of particular interest. The pathway was first approached using a process that started with the monomers and oxidized then. This was done because it was thought the radials formed during the oxidation reaction could potentially couple into dimers. Two lignans of coniferyl alcohol were chosen for analysis. By using these standards and comparing them to the beaker oxidation reactions of coniferyl alcohol in the presence of HRP and H2O2, these lignans could be identified in the oxidation products. This did not happen, but does not mean dimers did not exist in the products. The dimers chosen for analysis just were not present in the products. The pathway was then approached using a process that started with lignin, the larger structure, and attempted to break it down into dimers and monomers. This was done in an alkaline environment using NaOH. Results were inconclusive using this method, and stronger method development needs to be approached to determine the pathway of formation of the dimers from this angle.
Conclusion
Although some conclusions were made regarding the oxidation of lignin monomers, more research needs to be done to fully understand the oxidation process. Specifically, an analysis procedure needs to be determined for the products of the oxidation reaction. The oxidation reactions were successful, but no method was developed that allowed for successful analysis of the products of these reactions. Sinapic acid was also not oxidized, so a new approach needs to be done to oxidize this monomer and determine products of the reaction.
The EPR analysis of the monomers was successful as coupling constants were determined of the assumed radicals formed. This was only done with ferulic acid and coniferyl alcohol, so the other monomers need to be analyzed. It is likely they can be analyzed using the same method to determine these constants. It would be interesting to compare the acid and alcohol forms of the monomers for similarities in coupling constants. These constants can also then be compared among the monomers for similarities.
Little progress was made on the investigation into the pathway of formation of lignan. Two different approaches were used, but no conclusive results were developed for either of these. The degradation method presented the most promising results and literature is relatively abundant on the topic. More literature review should be done to determine a degradation method that can be completed and then analyzed on the HPLC. A successful method will assist with determining how lignan forms. This pathway can then be compared to the pathway of formation of lignin. Other lignans could also be researched to identify oxidation products of the monomers that could be dimers.
Future Directions
This current project did not reach any conclusions regarding the degradation of lignin. Although literature exists on this process, it was not able to duplicated in the lab. The next focus of this project involving lignin and lignan would be to determine the pathway that lignan forms. It was attempted to determine this pathway using a procedure that began with the monomers, but was unsuccessful in finding conclusive results. In response to these findings, it was decided to determine the pathway lignan forms by starting with the larger structure of lignin, and degrading it into monomers and dimers. More literature will need to be explored to determine a practical procedure to successfully degrade lignin and analyze it using HPLC. More lignan molecules could also be investigated using HPLC since a variety exist and are available for purchase from chemical companies. There are still many unknowns surrounding lignin and lignan and this project has the capability to be taken in many directions. The project will be taken over by Taylor Coffman with a focus on degradation methods as well as understanding the distinction between the lignin and lignan pathways of formation.
References
- ↑ Dimmel, D. (2010). Overview. Lignin and Lignans Advances in Chemistry. Taylor & Francis Group.
- ↑ Dimmel, D. (2010). Overview. Lignin and Lignans Advances in Chemistry. Taylor & Francis Group.
- ↑ Dimmel, D. (2010). Overview. Lignin and Lignans Advances in Chemistry. Taylor & Francis Group.
- ↑ Dimmel, D. (2010). Overview. Lignin and Lignans Advances in Chemistry. Taylor & Francis Group.
- ↑ Dimmel, D. (2010). Overview. Lignin and Lignans Advances in Chemistry. Taylor & Francis Group.
- ↑ Dimmel, D. (2010). Overview. Lignin and Lignans Advances in Chemistry. Taylor & Francis Group.
- ↑ Russel et al. (1996). Oxidative Coupling during Lignin Polymerization Is Determined by Unpaired Electron Delocalization within Parent Phenylpropanoid Radicals. Archives of Biochemistry and Biophysics, 332(2), 357-366.
- ↑ Russel et al. (1996). Oxidative Coupling during Lignin Polymerization Is Determined by Unpaired Electron Delocalization within Parent Phenylpropanoid Radicals. Archives of Biochemistry and Biophysics, 332(2), 357-366.
- ↑ Sturgeon, B. E., Chen, Y., & Mason, R. P. (2003). Immobilized Enzyme Electron Spin Resonance: A Method for Detecting Enzymatically Generated Transient Radicals. Analytical Chemistry, 75(19), 5006-5011.
- ↑ Kersten, P. J., Kalyanaraman, B., Hammel, K. E., Reinhammar, B., & Kirk, T. K. (1990). Comparison of lignin peroxidase, horseradish peroxidase and lactase in the oxidation of methoxybenzenes. Biochemical Journal, 268, 475-480.
- ↑ Kersten, P. J., Kalyanaraman, B., Hammel, K. E., Reinhammar, B., & Kirk, T. K. (1990). Comparison of lignin peroxidase, horseradish peroxidase and lactase in the oxidation of methoxybenzenes. Biochemical Journal, 268, 475-480.
- ↑ Sturgeon, B. E., Chen, Y., & Mason, R. P. (2003). Immobilized Enzyme Electron Spin Resonance: A Method for Detecting Enzymatically Generated Transient Radicals. Analytical Chemistry, 75(19), 5006-5011.
- ↑ Sturgeon, B. E., Chen, Y., & Mason, R. P. (2003). Immobilized Enzyme Electron Spin Resonance: A Method for Detecting Enzymatically Generated Transient Radicals. Analytical Chemistry, 75(19), 5006-5011.
- ↑ Sturgeon, B. E., Chen, Y., & Mason, R. P. (2003). Immobilized Enzyme Electron Spin Resonance: A Method for Detecting Enzymatically Generated Transient Radicals. Analytical Chemistry, 75(19), 5006-5011.
- ↑ Sturgeon, B. E., Chen, Y., & Mason, R. P. (2003). Immobilized Enzyme Electron Spin Resonance: A Method for Detecting Enzymatically Generated Transient Radicals. Analytical Chemistry, 75(19), 5006-5011.
- ↑ Dimmel, D. (2010). Overview. Lignin and Lignans Advances in Chemistry. Taylor & Francis Group.
- ↑ Lapierre, C. (2010). Determining Lignin Structure by Chemical Degradations. Lignin and Lignans Advances in Chemistry. Taylor & Francis Group.
- ↑ Dimmel, D. (2010). Overview. Lignin and Lignans Advances in Chemistry. Taylor & Francis Group.
- ↑ Lewis, N. G., Davin, L. B., & Sarkanen, S. (1998) Lignan and Lignan Biosynthesis: Distinctions and Reconciliations. American Chemical Society Symposium Series, 1-27.
- ↑ Rodríguez-García et al. (2019). Naturally Lignan-Rich Foods: A Dietary Tool for Health Promotion. Molecules, 24(917), 1-25.
- ↑ Warwick, K. (2020). How Healthful is Flaxseed? Medical News Today.
- ↑ Warwick, K. (2020). How Healthful is Flaxseed? Medical News Today.
- ↑ Kernan et al. (1997). Two New Lignans with Activity against Influenza Virus from the Medical Plant Rhinacanthus nasutus. Journal of Natural Products, 60(6), 635-637.
- ↑ Kernan et al. (1997). Two New Lignans with Activity against Influenza Virus from the Medical Plant Rhinacanthus nasutus. Journal of Natural Products, 60(6), 635-637.
- ↑ Rodríguez-García et al. (2019). Naturally Lignan-Rich Foods: A Dietary Tool for Health Promotion. Molecules, 24(917), 1-25.
- ↑ Rodríguez-García et al. (2019). Naturally Lignan-Rich Foods: A Dietary Tool for Health Promotion. Molecules, 24(917), 1-25.
- ↑ Warwick, K. (2020). How Healthful is Flaxseed? Medical News Today.
- ↑ Rodríguez-García et al. (2019). Naturally Lignan-Rich Foods: A Dietary Tool for Health Promotion. Molecules, 24(917), 1-25.
- ↑ Rodríguez-García et al. (2019). Naturally Lignan-Rich Foods: A Dietary Tool for Health Promotion. Molecules, 24(917), 1-25.
- ↑ Lewis, N. G., Davin, L. B., & Sarkanen, S. (1998) Lignan and Lignan Biosynthesis: Distinctions and Reconciliations. American Chemical Society Symposium Series, 1-27.
- ↑ Lewis, N. G., Davin, L. B., & Sarkanen, S. (1998) Lignan and Lignan Biosynthesis: Distinctions and Reconciliations. American Chemical Society Symposium Series, 1-27.
- ↑ Lewis, N. G., Davin, L. B., & Sarkanen, S. (1998) Lignan and Lignan Biosynthesis: Distinctions and Reconciliations. American Chemical Society Symposium Series, 1-27.
- ↑ Lewis, N. G., Davin, L. B., & Sarkanen, S. (1998) Lignan and Lignan Biosynthesis: Distinctions and Reconciliations. American Chemical Society Symposium Series, 1-27.
- ↑ Lewis, N. G., Davin, L. B., & Sarkanen, S. (1998) Lignan and Lignan Biosynthesis: Distinctions and Reconciliations. American Chemical Society Symposium Series, 1-27.
- ↑ Rodríguez-García et al. (2019). Naturally Lignan-Rich Foods: A Dietary Tool for Health Promotion. Molecules, 24(917), 1-25.
- ↑ Lapierre, C. (2010). Determining Lignin Structure by Chemical Degradations. Lignin and Lignans Advances in Chemistry. Taylor & Francis Group.
- ↑ Lapierre, C. (2010). Determining Lignin Structure by Chemical Degradations. Lignin and Lignans Advances in Chemistry. Taylor & Francis Group.
- ↑ Lapierre, C. (2010). Determining Lignin Structure by Chemical Degradations. Lignin and Lignans Advances in Chemistry. Taylor & Francis Group.
- ↑ Lapierre, C. (2010). Determining Lignin Structure by Chemical Degradations. Lignin and Lignans Advances in Chemistry. Taylor & Francis Group.
Personal Reflection: Progress and Knowledge
Before I first started doing research with Stephanie Saey as a sophomore, I was under the impression you mixed compounds together and hoped for the best for research. My eyes were opened when I realized much more goes into research. In fact, more time is spent outside of the lab planning and analyzing than actually being in the lab. I learned method development is an important aspect of research. You need to go into the lab with a plan, and an alternate plan sometimes. My research experience before coming to Monmouth was so juvenile. I did science fair presentations on such silly things that I thought were so revolutionary and life changing at the time. I look back on my research here and think, I really did make the most out of my experience. I accomplished more than I ever thought I would, and I enjoyed my research here. Yes at times it was a pain, and I put it on the back burner, but I always had a passion for my research. I guess you could say I'm a lignin superfan now! I think greater growth, both as a student and as a researcher, occurred through my research failures. At first, failures felt like a slap in the face. But as my experience progressed, I realized these failures can be turned into successes. Failures mean you have successfully figured out something about your research doesn't work. It can guide you or direct you to the next step in your research. These "failures" pushed me to work harder and crave to find the explanation for different things I was interested in. I think being able to see this project grow from the start really cultivated my interest and love for this project. I have this curious and stubborn drive within me that continues to push me toward my goals and aspirations. The mentality I have leaving this research is completely opposite the mentality I had beginning it! Although there were some bumps along the way, I wouldn't change one thing! I have growth throughout this research, for the better, and have hopefully set it up well for the next person to take over. My experience here has been filled with my failures, but not once did I give up and lose sight at the finish.
Success is the ability to go from one failure to another with no loss of enthusiasm – Winston Churchill