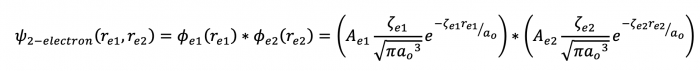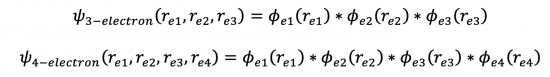PCh10 lec 2
(4/27/20, bes)
Happy last full week of classes!
So, here are some thoughts:
- 1) You should have received your exam 2 score along with a link to an exam 2 summary page. Like with all exams, you should review the exam...in this case by looking over your exam and the summary page to figure out what material you did not fully understand. In a best case scenario, i would "hand back" your exams...the notes i wrote on your exam will assist you in identifying your errors...but since i am working at home and do not have a commercial, multi-document scanner i did not plan to return graded exams until you juniors returned to campus (sorry seniors). [NOTE on grading...as a professional educator, i allocate my time to maximize learning...so, when grading an exam that will not be returned, i do not allocate the time to writing correct answers/notes on exams since you will not see them. This is often done with final exams that are not returned. So, when or if you get your exams back you might be disappointed that i did not make notes (did not fully identify your errors).] Let me also remind you that since the last exam covers only Chapter 9, 10, 15...you will not need to review the exam 2 material for a comprehensive final.
- 2) Allocation of my time...under normal curcumstances, it takes me very little time to prepare for Gen Chem lectures; Gen Chem labs have been refined and Steve does nearly all of the setup required. PChem (and research) students benefit from this since i normally have more time to allocate to them. Under these online conditions, i am having to allocate a large portion of my time to generating online content for lecture and lab, hence less time for you...and pchem in general. I look forward to returning to campus and returning to "normal"...i hope you do too. Although this statement of allocation of time seems unrelated to quantum mechanics...it is truly a key aspect that i will share at a later time...
Many-Electron Wavefunctions
(Sec 10.1 - 10.3) <--but i will not cover all of this now...
So, as noted, the multi-electron Schrodinger equation is quite complex and there is no analytical solution. <-- this term analytical solution is worth expanding on for a minute...
Analytical Solution vs Numerical Solution
- An analytical solution is a mathematical function/equation that is a solution to a differential equation (or any operator equation). For example, we have developed/solved the functions/equation for the hydrogen atom back in chapter 9. These functions can be plotted in Mathematica or Igor or Excel.
- A numerical solution is a set of numbers, usually (x, y) data set that would look very similar to data that comes off of the HPLC, UV-Vis, or IR. It is often the case that a set of numbers may be approximated by a mathematical function/equation but this DOES NOT make it an analytical solution.
So, to continue with the multi-electron Schrodinger equation...and in particular...solving the multi-electron Schrodinger equation...we must figure out a numerical solution to this equation...this is how we do it:
- - We know the 1-electron wavefunctions; it is not too crazy to expect that these functions are similar to the 2-electron wavefunctions. So, these 1-electron wavefunction will be the foundation of our numerical solutions...
- So if there are 2 electrons, why not use the 1s orbital for the first electron and the 2s orbital for the second electron? This would minimize the interaction between the 2 electrons and might be a good answer. Now you all know this that is not correct since you have been taught, but not explained why, that there are 2 electrons per orbital. I will remind you that we skipped chapter 6. I feel that this material is a bit randomly presented and is not really needed until NOW...feel free to skim chapter 6, but i will guide you thru the most important parts here...ie the Stern- Gerlach Experiment...watch this 3 min video: Stern-Gerlach Experiment (U2 07 03)
- The result from the Stern-Gerlach experiment is that the electron has something inherent to it and we call this something electron spin. Now if you like watching videos, the following one goes into a little bit more details (there are many others)...the take home message here is that electrons have an intrinsic property called "spin" and an electron can have 2 values for spin...spin up or spin down...or ms = +1/2 and ms=-1/2. With the final take home point being that you can have 2 electrons in each orbital and using the 4th quantum number (spin quantum number), ms we can specify exactly each electron.
- What is Spin? | Quantum Mechanics <-- optional 10 min video
Atomic Orbital Approximation
When we use the hydrogen atom atomic orbitals to assist in generating a numerical solution to the multi-electron Schrodinger equation, we refer to this as the atomic orbital approximation. Note the the 1s orbital wave function has the nuclear charge (Z) in this term, but since it is = 1, then we don't "see" it.
The way that we adjust this H-atom atomic orbital wavefunction is first to change the Z into a ζ (a zeta)...
Back to the 2 electron system...so due to electron spin we find that there are 2 electrons per orbital each with their own set of quantum numbers (n, l, ml, ms) and due to the electron-electron repulsion, this has an end result of each electron not "feeling" the entire nuclear charge of 2...Z = 2, but ζ < 2. The 1s orbital in a 2-electron system is slightly "expanded" due to the nuclear charge acting on 2 electrons as opposed to just one in the H-atom...this is not a bad initial approximation.
So what might a 3-electron or a 4-electron system look like?
- where the additional wavefunctions will use the H-atom 2s orbitals with a ζ for each of these electrons...and the general form of the obrital approximation would look like this:
END OF LECTURE.