Homolytic Bond Dissociation Energies
Homolytic bond dissociation energies or just bond dissociation energies (BDE) is a measure of a particular bond strength. BDE are also referred to as bond enthalpies.
Example
For example, the BDE for the C-C bond in
- H3C-CH3 --> H3C• + •CH3
can be determined using the calculated enthalpies of formation (ΔHf) using the following method:
HF/6-31(G)d
Using WebMO/Gaussian to calculate the ΔHf(H3C-CH3), returns the following:
The RHF (Restricted HF) energy, ie. ΔHf(H3C-CH3) is reported in units of Hartree, -79.2287548119 Hartree
Using WebMO/Gaussian to calculate the ΔHf(CH3•), returns the following:
The RHF (Restricted HF) energy, ie ΔHf(CH3•) is reported in units of Hartree, -39.5589916118 Hartree
- ΔHrxn = ΔHf(Products) - ΔHf(Reactants)
- ΔHrxn = 2*ΔHf(CH3•) - Hf(H3C-CH3)
- ΔHrxn = 2*-39.5589916118 - (-79.2287548119)
- ΔHrxn = 0.110771588 Hartree = 290.830826448 kJ/mol
- According to Engel, 2nd, Chapter 15, table 15.2, the actual value is 406 kJ/mol)
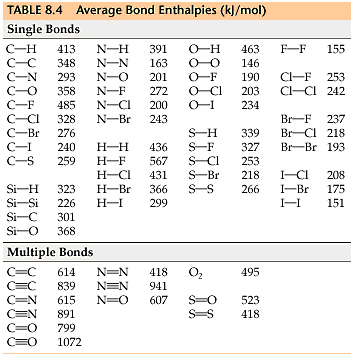
As can bee seen in this table (shown in all Gen Chem textbooks), the "average bond enthalpy" for a C-C bond is 348 kJ/mol.
B3LYP/6-311+G(2d,p)
Using a more accurate computational approach, B3LYP/6-311+G(2d,p):
- - B3LYP Energy -39.8561207613 Hartree
- - B3LYP Energy -79.8583705307 Hartree
- ΔHrxn = 0.14613 Hartree = 384 kJ/mol
