Andrew S Ferris
Personal Information
Senior Biochemistry Major
Hometown: Galesburg, IL
Undergraduate Research Activities
Summer 2019
Kieft Summer Research with Bradley Sturgeon (Electron Paramagentic Resonance)
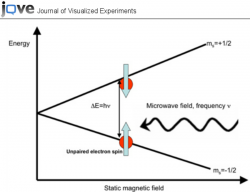
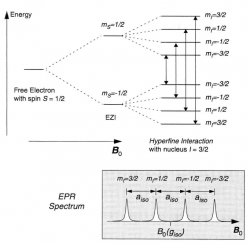
Electron Paramagnetic Resonance (EPR) is procedure which utilizes the the splitting in energy levels between the two possible spin configuration of an electron in the presence of a magnetic field, in order to detect the coupling of a given electron to the nuclei of an adjacent atom. Once the substance is introduced to a magnetic field of a given strength, a range of microwave frequencies is panned through in order to find the frequency with the resonant energy. An unpaired electron which is delocalized evenly throughout a molecule will only show one iteration of splitting, the splitting in energy levels between the two spin configurations caused by the magnetic field. However, in a system where additional interactions exist between magnetic field created by the electron and the nuclear magnetic moment of a given atomic nuclei, Hyperfine Splitting will occur. This further splitting infers that the unpaired electron is not evenly delocalized around the all nuclei in the molecule, rather it is interaction more strongly at some positions (atomic nuclei) in the molecule than others. The phenomena is called hyperfine splitting becuase it further splits the primary splitting caused by the magnetic field. However, since the biased interactions between the electron and a given nuclei is quite minute, the splitting caused by these interactions also pale in comparison to to the original splitting.
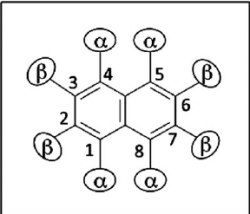
Hyperfine Splitting provides information on the magnitude of the electron's interaction with a given atomic nuclei when the electron is not evenly delocalized. The magnitude of the splitting is proportional to the strength of the electron-nuclei interactions and therefor the amount of time the electron spends in that position (a evenly delocalzied electron would spend the same amount of time on all positions and therefore no hyperfine coupling would be present). The magnitude of the splitting is recorded in energy unit of Gauss, and is displayed on the EPR spectrum as the distance between peaks. Quantitatively the magnitude of the hyperfine splitting (and therefore the strength of the ineraction between the eloectron and that given atomic nuclei) is represented by the Hyperfine Coupling Coefficient. An unpaired electron was transferred to naphthalene by Sodium metal, via a reduction reaction. Where a molecule such as Benzene would have an unpaired electron evenly delocalized around the entire structure due to its chiral symmetry, a molecule such as Naphthalene would have two different positions due to its chiral asymmetry. We would expect to see two different Hyperfine Coupling Coefficients, one for each unique position.