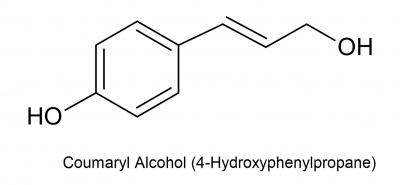
Preparation of coumaryl alcohol
Revision as of 06:24, 13 October 2021 by Sampsonyte4 (talk | contribs)
Joseph Sampson, 10/21/21
This is a continuation of a project started in 2018 by A. Tibbs.
Coumaryl alcohol is a monomer of lignin.
Coumaryl alcohol will be synthesized from coumaric acid through two separate reactions:
- First, coumaric acid will be converted into ethyl coumarate through esterification. This will involve converting the carboxylic acid group into an ethyl ester group.
- Finally, ethyl coumarate will be converted into coumaryl alcohol through a reduction reaction.
The experiment performed is based off of a published paper in which the authors previously synthesized coumaryl alcohol. The "Synthesis of p-Coumaryl Alcohol" subheading is what's important.
- Initial procedure for the Synthesis of Coumaryl Alcohol (simplified):
- Add 1g (0.0061 moles) of p-Coumaric acid and 5mL (0.07 moles) acetyl chloride into a round bottom flask containing 50mL of dry ethanol. The total concentrations of coumaric acid and acetyl chloride in solution should be 0.111M and 1.27M each.
- Stir at room temperature for 17 hours.
- After 17 hours, evaporate the solvent.
- Repeat steps 1-3 again.
- Recrystallize the product from ethyl acetate (product should be ethyl p-coumarate).
- Add 640mg of ethyl coumarate to 30mL of toluene and place this solution on ice and under nitrogen.
- To the mixture on ice and under nitrogen, SLOWLY add 12mL of DIBAL-H.
- Stir for 1hr while still ice-cooling and under nitrogen.
- Add 5mL ethanol to quench the reaction and then mix some more.
- Partially remove the solvent under reduced pressure (???)
- Add 50mL water and mix.
- Transfer mixture to separatory funnel and add ethyl acetate, shake it up, and then extract the aqueous layer. The organic layer contains coumaryl alcohol [don’t lose any of it!!].
- Repeat step 12 three more times (4 extractions total).
- Recrystallize the organic layer (coumaryl alcohol) from dichloromethane. The product should look like pale yellow crystals.