Acetaminophen Radicals
The Ab-initio computation of the acetaminophen radical. The goal of this project is to elucidate where electrons are located on the molecule when a radicalization occurs.
Background
Acetaminophen(APAP) is an active ingredient in many over-the-counter and prescription painkillers, such as Tylenol and Oxycodone. APAP is also responsible for approximately 50% of the cases of acute liver-failure in the United States and Great Britain. Treatments for acetaminophen-induced liver injury(AILI) are limited. The current mechanism for AILI is the production of hepatotoxic NAPQI as a metabolite in an enzymatic, two-electron oxidation. However, evidence also supports a one electron oxidation.
Web MO/Gaussian
Web MO/Gaussian is a software containing various basis sets that allow for various levels of computational chemistry.
B3YLP/6-311+G(2d,p)
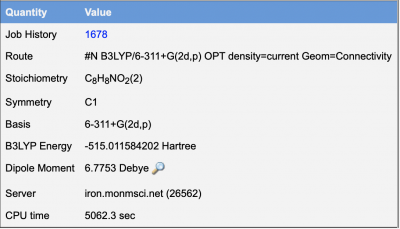
After running a calculation on Web MO/Gaussian, the following results were reported.
Analysis
Radical Location
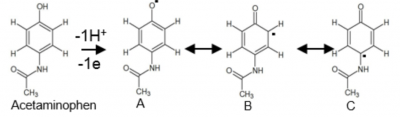
As seen on the electron density map above, the radical spends a lot of time at the ortho positions. To aid with this, the possible resonance structures are shown below.
Knowing this information is important because it helps with predicting which dimer products will form. Certain dimers may be biologically active, which could play a role in hepatotoxicity.
ΔHf Calculations
Below are the calculations of the ΔHf for the formation of a radical of the phenoxyl in acetaminophen. The values listed in Hartree were found doing geometry optimizations using B3LYP/6-311+G(2d,p) basis sets.
- ΔHrxn = ΔHf(Products) - ΔHf(Reactants)
- ΔHrxn = ΔHf(-OH•) - Hf(-OH)
- ΔHrxn = (-515.01158420 Hartree) - (-515.480291398 Hartree)
- ΔHrxn = 0.468707198 Hartree = 1230.59084209 kJ/mol
Cites
This paper has ESR data on the acetaminophen radical https://ehp.niehs.nih.gov/doi/pdf/10.1289/ehp.8564127