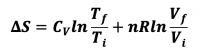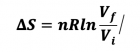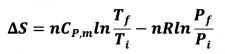PChem312 f20 w13
Monday, Nov 9, 2020
We will now highlight how to calculate entropy (ΔS) for four processes involving ideal gases:
- 1) Reversible Isothermal Expansion or Compression
- ViTi --> VfTi
- 2) Constant P, V temperature change
- ViTi --> ViTf
- PiTi --> PiTf
- 3) Changes in both V and T, constant P
- ViTi -->V fTf
- 4) Changes in both P and T, constant V
- ViTi -->V fTf
Example Problem 1
- (Please write out your complete solution and email to bes by next class period)
Consider a gas undergoing the following change under constant pressure (1.00 bar) conditions; calculate the change in entropy (ΔS): Vi = 10.0 L; Ti = 298 K; Tf = 398 K
Example Problem 2
- (Please write out your complete solution and email to bes by next class period)
Consider a gas undergoing the following change under constant volume (10.0 L) conditions; calculate the change in entropy (ΔS): Pi = 1.00 bar; Ti = 298 K; Tf = 398 K
Wednesday, Nov 11, 2020
- Review contributions to ΔS due to ΔT, ΔV, ΔP for ideal gas/closed system.
- Entropy of Phase Changes...
...continuing numbering from above...
- 5) Solid --> Liquid phase change (fusion/melting/freezing)
- - under constant pressure
- - under constant volume
- 6) Liquid --> gas phase change (vaporization/boiling/condensation)
- - under constant pressure
- - under constant volume
Example Problem 3
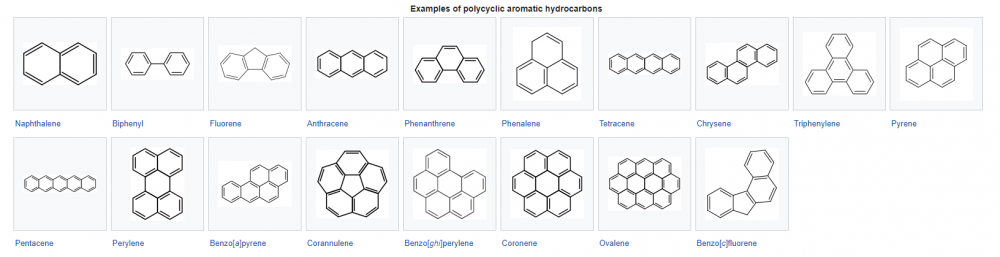
Poly aromatic hydrocarbons (<-- wiki link here) represent and interesting set of compounds.
Calculate the entropy of fusion for 5 PAHs.