Ian Salveson: Research Log
June 15, 2016
Determine the effect of flow rate on product formation. Figure 1 below shows similar reactions with differing flow rates.
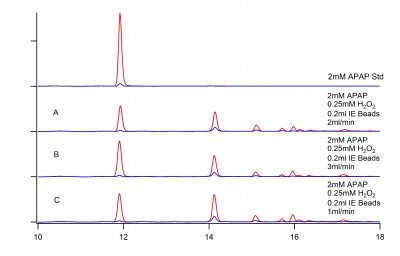
June 16, 2016
Concentration of acetaminophen is increased in preparation for Flash Chromatography. Figure 1 below shows the effect of increasing concentration on product formation.

It appears that at higher concentrations, the reaction will begin to form polymeric material, despite relatively low concentrations of Hydrogen Peroxide.
Sample B will be separated using Flash Chromatography.
It is important to note that we have reached the max absorbance of our instrument.
Figure 2 below shows a set of samples under identical conditions as Figure 1 except with a flow rate of 2 ml/min.
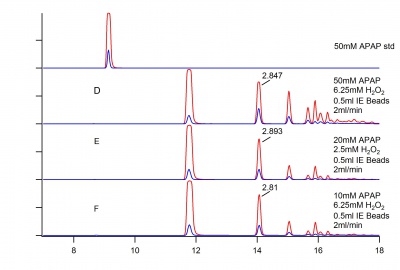
The chromatography on this figure is faulty, specifically the changing retention time. The results also contradict what may have been expected of a faster flow rate. These samples should be run again if the data is deemed significant.