Lignin Monomers: Oxidation and Degradation
Abstract
The lignin polymer is synthesized via oxidative coupling of three basic monomers: p-coumaryl alcohol, coniferyl alcohol, and sinapyl alcohol. These three monomers each possess a phenol group that stabilizes the radical intermediate generated during the oxidation reaction. Although some literature exists showing the ESR spectra of these short-lived radical intermediates, we have revisited these intermediates using a combination of experimental techniques [HPLC and immobilized enzyme-ESR technique (IE-ESR)] and computational approaches [B3YLP/ESR-II and WINSIM]. Two radicals dimerize together to create lignan, which is biologically active and currently being tested for pharmacological properties. HPLC was used to study the oxidation of coniferyl alcohol, the monomeric lignan, and validate the formation of dimers. The mechanisms of this dimer formation were also studied to better understand the oxidative coupling of the lignin monomers.
Introduction
COMING SOON
Lignin
Lignan
Lignan is formed from the oxidative coupling of two lignin monomers. The dimers are often linked between the beta-carbons on the side chain of each monomer. Many of the dimers stated often in literature possess this linking structure. These dimers have biological activity, whereas the larger structure of lignin does not. This allows them to have potential healths benefits associated with them. Some of these benefits are anti-mubial, anti-inflammatory, and antioxidant. The relationship between allergies and lignan has also been studied. It might be related to having an anti-histamine effect on allergy symptoms.
Research is currently being done to study the relationship between cancer and lignan. Breast cancer has been studied the most, but other cancers such as prostate and ovarian are being researched. Currently there are no conclusions regarding those cancers, as the research is in beginning stages. Many studies are centered around whether consumption of flax seeds correlates to a reduction in the risk of breast cancer, as well as the mortality. The reason flax seeds are being researched is because they have the highest concentration of lignans. With breast cancer, it was found flax seed consumption did decrease the risk of breast cancer, as well as the mortality of breast cancer, in post-menopausal women. This is believed to be because the lignan acts as a competitive inhibitor to estrogen. Some lignans, such as matairesinol and lariciresinol, have structures similar to estrogen. This allows them to bind to the estrogen receptor on the cancer cell, therefore blocking estrogen from binding. This prevents further growth of the cancer.
Experimental Methods
Oxidation of Lignin Monomers
Beaker reactions with varying reaction conditions were completed to analyze the oxidation of each monomer. For each monomer, 100 mL of a 2 mM standard solution of the monomer (purchased from Sigma Aldrich) was made using 50/50 dioxane/pH 5 buffer. The pH 5 buffer was a phosphate-citrate buffer (purchased from Sigma Aldrich). Three reactions were then completed with varying concentrations of hydrogen peroxide in the presence of a constant HRP concentration in scintillation vials. The first reaction was composed of 5 mL of the 2 mM substrate, 10 μL of 0.5 M hydrogen peroxide (1 mM final concentration hydrogen peroxide) and 5 μL of HRP. The second reaction was composed of 5 mL of the 2 mM substrate, 5 μL of 0.5 M hydrogen peroxide (0.5 mM final concentration hydrogen peroxide) and 5 μL of HRP. The third reaction was composed of 5 mL of the 2 mM substrate, 5 μL of 0.5 M hydrogen peroxide (0.25 mM final concentration hydrogen peroxide) and 5 μL of HRP. These reactions were then analyzed on the HPLC for oxidation products of each monomer and compared.
Analysis of Monomer Oxidation
The reactions and the standards were analyzed on the HPLC using an acetonitrile (ACN) and water gradient for 25 minutes. The first 10 minutes were ran at 100% water, 10-20 minutes were ran at an 80% ACN/20% water gradient, and the remaining 5 minutes at 100% water.
Immobilization of Enzymes
An immobilization of peroxidase from horseradish (HRP) was completed using Affi-Gel® 10. This procedure is centered around the isolectric point of the enzyme. The immobilization was completed using a MOPS buffer (pH 7.0) and about 1 mL of Affi-Gel® 10 beads.
Setup of Enzyme Immobilization
Made 0.190 mM immobilized enzyme solution (abs 1.94) using a simple apparatus. A Poly-Prep® Chromatography Column (Bio-Rad Laboratories, Inc. product 731-1550) and 3-way stopcock (Bio-Rad Laboratories, Inc. product 732-8103) were used. The column was secured to a ring stand and the 3-way stopper was attached. A 5 mL lure-lock syringe was attached to the 3-way stopcock at a 90° angle. The initial HRP solution is a 5 mM solution of peroxidase from horseradish and MOPS buffer. About 1 mL of Affi-Gel® 10 beads were added to the column attached to the ring stand and washed with water. They were then washed with MOPS buffer. The peroxidase and buffer solution in the eppendorf tube was added to the column containing the Affi-Gel® 10 beads and mixed by inverting and flicking the bottom of the column. The column was sealed and placed on a LabQuake® for an hour.
Oxidation of Lignin Monomers Using Immobilized HRP
Beaker reactions with immobilized HRP
Beaker reactions were completed using the immobilized HRP and analyzed on the HPLC. The reactions had varying concentrations of hydrogen peroxide. A 2 mM standard was made for each monomer with a volume of 100 using 50/50 dioxane/pH 5 buffer. Before the immobilized HRP was used, it was rinsed twice with MOPS buffer. The first reaction was composed of 5 mL of substrate, 10 µL of 0.5 M hydrogen peroxide (1 mM final concentration hydrogen peroxide), and 50 µL of immobilized HRP. The second reaction was composed of 5 mL of substrate, 5 µL of 0.5 M hydrogen peroxide (0.5 mM final concentration hydrogen peroxide), and 50 µL of immobilized HRP. The third reaction was composed of 5 mL of substrate, 2.5 µL of 0.5 M hydrogen peroxide (0.25 mM final concentration hydrogen peroxide), and 50 µL of immobilized HRP. These reactions were then analyzed on the HPLC for possible oxidation products. The results were then compared to previously collected HPLC data using HRP, opposed to immobilized HRP.
Flat Cell Reactions with Immobilized Enzyme
A flat cell was loaded with glass wool, sand, and immobilized enzyme. Glass wool was lightly packed into the bottom neck of the flat cell. A small amount of sand was then mixed with nanopure water and added into the flat cell about 2 mm above the glass wool using a plastic 1 mL syringe. About 50 μL of immobilized enzyme is added on top of the glass wool and sand to fill most of the cavity of the flat cell. The pH 5 buffer was ran through the flat cell for about 10 minutes. A 1 mM ferulic acid, 1 mM hydrogen peroxide solution in 50/50 dioxane/pH 5 buffer was made. This solution was put in a 60 mL plastic syringe and flow controlled through the flat cell at 0.25 mL/minute, 0.5 mL/minute, 1 mL/minute, and 2 mL/minute. Flow was collected for each rate and analyzed on the HPLC. The same procedure was completed for p-coumaric acid.
EPR Analysis with Immobilized Enzyme
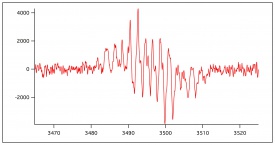
A flat cell was loaded with glass wool, sand, and immobilized enzyme using the procedure outlined above. A 10 mM ferulic acid solution in 50/50 dioxane/pH 7.4 buffer was made. Hydrogen peroxide was added to a concentration of 10 mM before EPR analysis. The solution was placed in a 60 mL plastic syringe and flow was controlled. Flow was analyzed at rates of 0.5 mL/minute, 1 mL/minute, and 2 mL/minute. The parameters for the program were modulation amplitude 0.5 G, sweep width 40.0 G, and center field at 3492.0 G.
Lignan Beaker Reactions
Beaker reactions of lignans were done with varying concentrations of hydrogen peroxide. Two lignan standards, (+)-pinoresinol and matiaresinol (purchased from Cayman Chemical, of 9.3 mM were created. A 2 mM coniferyl alcohol standard in 50/50 dioxane/pH 5 buffer was made. The first reaction was composed of 5 mL of substrate, 10 µL of 0.5 M hydrogen peroxide (1 mM final concentration hydrogen peroxide), and 5 µL of HRP. The second reaction was composed of 5 mL of substrate, 5 µL of 0.5 M hydrogen peroxide (0.5 mM final concentration hydrogen peroxide), and 5 µL of HRP. The third reaction was composed of 5 mL of substrate, 2.5 µL of 0.5 M hydrogen peroxide (0.25 mM final concentration hydrogen peroxide), and 5 µL of HRP. These reactions were then analyzed on the HPLC for lignan identification.
Lignin Degradation Using Alkaline Conditions
Lignin (purchased from Sigma Aldrich) was degraded in an alkaline solution of 100 mM NaOH. A solution of 50 grams of lignin and 10 mL of 100 mM NaOH were stirred on a stir plate for 24 hours. Samples of 10 µL of the mixture were extracted at 1 minute, 24 hours, and 48 hours after stirring and placed in 3 mL of pH 5 buffer. The samples were analyzed on the HPLC for degradation products.
Notes of Safety
Dioxane
Molecular Formula: C4H8O2
Molecular weight: 88.11 g/mol
Physical appearance: clear, colorless liquid
1,4-dioxane has the potential to be an eye, skin, and digestive tract irritant. To avoid contact, lab goggles, gloves, and protective clothing should be worn. In the case of exposure, eyes and skin should be flushed with water. If ingested, vomiting should not be induced and medical aid should be sought. Keep away from heat, sparks, and flame and use in a fume hood. Inhalation could induce breathing difficulties. It should be stored under nitrogen to less peroxide formation. If it is noticed that peroxide formation has occurred, do not open the container unless a professional. It has the potential to form explosive peroxides with prolonged exposure.
Sodium hydroxide
Molecular formula: NaOH
Molecular weight: 40 g/mol
Physical appearance: solid, white
Sodium hydroxide can cause eye, skin, and digestive tract burns. In the case of contact with sodium hydroxide, flush the eyes and skin with water for at least 15 minutes and seek medical attention. Vomiting should not be induced, but water can be drank. To mitigate these risks, lab safety googles, gloves, and protective clothing should be worn. Sodium hydroxide is hygroscopic so it must be stored in a tightly closed container. It is corrosive to material so caution is needed in case of a spill. Material should be disposed of in the proper disposal container.
Results
Lignin
HPLC Analysis
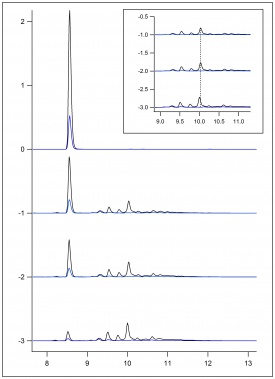
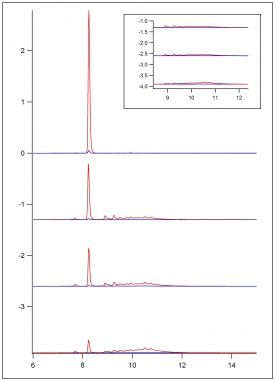
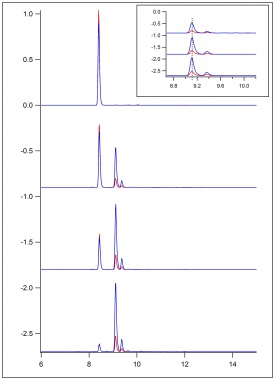
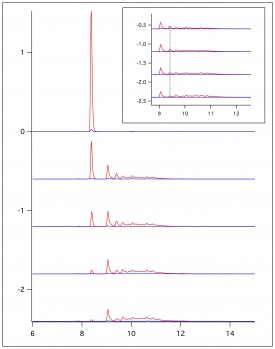
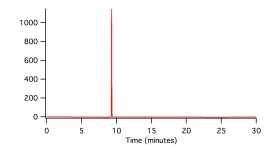
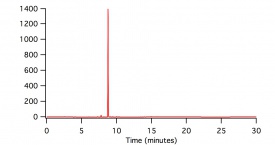
In figure 1, figure 2, and figure 3, beaker reactions of the oxidation of the three lignin monomers using H2O2 was completed. The same procedure was done using a 2 mM solution of the standard in 50/50 dioxane and pH 5 buffer. Dioxane was introduced into the reaction to assist with possible product formation solubility. Each reaction consisted of 5 mL of substrate, 5 μL of HRP, and a varying amount of 0.5 M H2O2 to reach final concentrations of 0.25 mM, 0.5 mM, and 1.0 mM. In figure 1 and figure 2, a standard is being consumed and producing oxidation products. These products could be dimers or small polymers. As the concentration of H2O2 increases, the more the standard is consumed and the more products formed. Since each reaction only differed by the concentration of the H2O2, each reaction can be compared. In figure 3 sinapic acid did not produce product peaks because the standard was never consumed. By changing the substrate to only being in pH 5 buffer, product peaks were seen.
In figure 4, figure 5, and figure 6, the same beaker reactions were completed as above using immobilized HRP in agarose beads. HPLC analysis was completed to ensure the enzyme was immobilized on the beads. Reactions were also completed to show reproducibility of previously collected HPLC data of the monomers. These reactions used 5 mL of substrate, 5 μL of HRPi, and a varying amount of 0.5 M H2O2 to reach final concentrations of 0.25 mM, 0.5 mM, and 1.0 mM. These figures showed the standard being decreased and oxidation products being formed. As the concentration of the H2O2 increased, more standard was consumed and more oxidation products were formed. Figure 6 is showing sinapic acid producing product peaks, but no peaks were shown in figure 3 with sinapic acid. This is because the substrate was changed to only be in pH 5 buffer opposed to 50/50 dioxane and pH 5 buffer.
To evaluate the use of the immobilized enzyme in a flat cell, samples of the waste were collected to ensure the immobilized enzyme was functioning in the flat cell. To do this, a flat cell was loaded with steel wool, sand, and agarose beads with HRPi. MOPS Buffer was then ran over the beads in the flat cell to wash them and ensure they were not dry. A substrate of 1 mM ferulic acid and 1 mM hydrogen peroxide in 50/50 dioxane/pH 5 buffer was then ran over the immobilized enzyme in the flat cell. The flow of the substrate was controlled using a syringe pump at 0.25 mL/min, 0.5 mL/min, 1 mL/min, and 2 mL/min. Samples of waste were then collected at each flow rate and analyzed on the HPLC. The results of ferulic acid are shown in figure 7. The same procedure was used for a substrate of 1 mM p-coumaric acid and 1 mM hydrogen peroxide in 50/50 dioxane/pH 5 buffer and is shown in figure 8.
EPR Analysis
Figure 9 shows previously collected EPR data of the oxidation of lignin monomers. In figure 10, figure 11, figure 12 and figure 13, preliminary ESR data is shown To complete preliminary ESR data collection using ferulic acid, a substrate of 1 mM ferulic acid in 50/50 dioxane/pH 5 buffer was made. The flat cell was then loaded with steel wool, sand, and immobilized enzyme. Buffer was ran over the immobilized enzyme for about an hour. The flat cell was then mounted in the ESR. Hydrogen peroxide was added to the substrate directly before flow was began so its concentration in solution was 1 mM. The substrate was then flowed through the flat cell in the ESR at a flow rate of 1 mL/min and a signal was collected. The flow of the substrate was then increased to 2 mL/min and a signal was collected. The flow of the substrate was then stopped, and no signal was apparent. This was done to ensure the signal being produced was a result of the oxidation of the monomers. If this was true, stopping the flow would stop the signal. The flow was then re-started with a flow rate of 2 mL/min, and the signal returned.
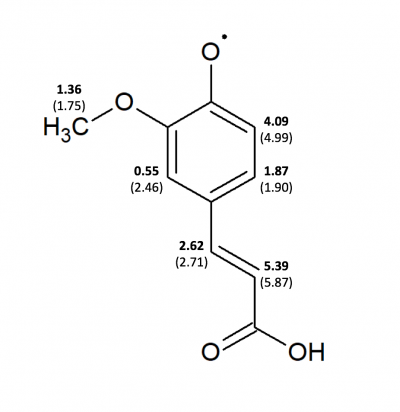
Computational analyses were completed of ESR experimental data of ferulic acid and coniferyl alcohol. A computational program called WINSIM was used to compare experimental data with simulated data. Simulating the experimental data allowed for the determination of coupling constants of the assumed radicals formed. This allowed determination of which radicals were more likely to form and potentially couple together.
Computational Analysis
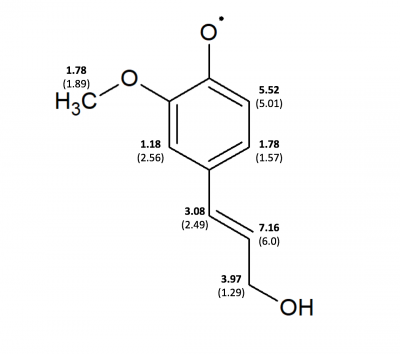
|
| Position | Coniferyl Alcohol (G03) | Coniferyl Alcohol (sim-Burrkitt) Chemical ox | Coniferyl Alcohol (sim) g-shift -0.26, LS 22, LW 0.32 | Ferulic Acid (G03) | Ferulic Acid (sim-Burkitt) | Ferulic Acid (sim) g-shift: 1.46, LS: 80.9, LW: 0.34 |
| 10 (ortho to -OH) | 5.01 | 5.33 | 5.52 | 4.99 | 5.56 | 4.09 |
| 11 (meta to -OH) | 1.57 | 1.86 | 1.78 | 1.90 | 1.70 | 1.87 |
| 20 (ortho to -OCH3) | 2.56 | 1.16 | 1.18 | 2.46 | 1.53 | 0.55 |
| 19 (on alpha C | 2.49 | 2.32 | 3.08 | 2.71 | 2.64 | 2.62 |
| 18 (on beta C) | 6.0 | 5.50 | 7.16 | 5.87 | 5.28 | 5.39 |
| 17 (on gamma C | 1.29 | 3.92 | 3.97 | x | x | x |
| 21 (-OCH3) | 2.77 | 1.38 | 1.78 | 0.15 | 1.69 | 1.36 |
| 22 (-OCH3) | 0.15 | 1.38 | 1.78 | 2.54 | 1.69 | 1.36 |
| 23 (-OCH3) | 2.75 | 1.38 | 1.78 | 2.55 | 1.69 | 1.36 |
Lignan
HPLC Analysis
Discussion
Conclusion
Future Directions
This current project did not reach any conclusions regarding the degradation of lignin. Although literature exists on this process, it was not able to duplicated in the lab. The next focus of this project involving lignin and lignan would be to determine the pathway that lignan forms. It was attempted to determine this pathway using a procedure that began with the monomers, but was unsuccessful in finding conclusive results. In response to these findings, it was decided to determine the pathway lignan forms by starting with the larger structure of lignin, and degrading it into monomers and dimers. More literature will need to be explored to determine a practical procedure to successfully degrade lignin and analyze it using HPLC. More lignan molecules could also be investigated using HPLC since a variety exist and are available for purchase from chemical companies. There are still many unknowns surrounding lignin and lignan and this project has the capability to be taken in many directions. The project will be taken over by Taylor Coffman with a focus on degradation methods as well as understanding the distinction between the lignin and lignan pathways of formation.