N-Acetyl L-Tyrosine
Revision as of 17:26, 29 June 2016 by Benstillwell (talk | contribs)
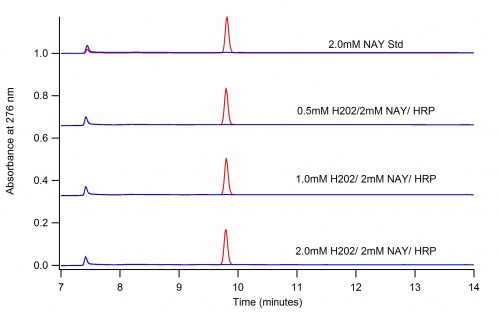
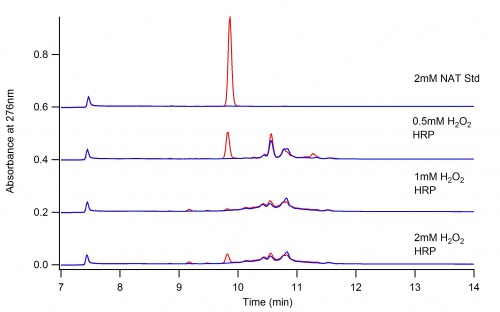
This page depicts the oxidation of N-Acetyl L-Tyrosine (NAT) by Horse Radish Peroxidase (HRP)
The Dioxane Conundrum
The following two graphs depict the difference in reaction when dioxane is present and when it is not.
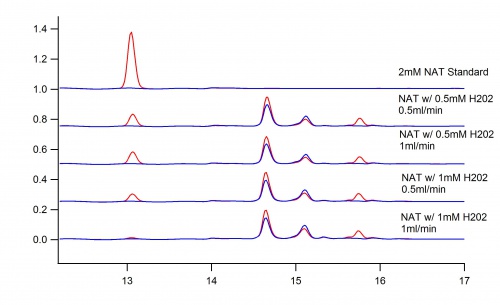
Beaker Reaction vs. Immobilized Enzyme Bio-Reactor
Utilizing the immobilization technique in a bio-reactor allows for the control of the rate of flow of the reaction. This makes the peaks in the products much more defined.
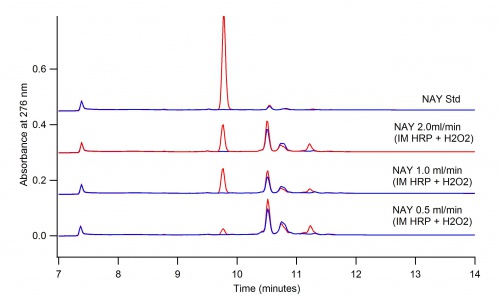
Figure 3: 2mM NAT oxidation without dioxane and reacted with 1mM H2O2 using the Immobilized Enzyme technique 6/9/16