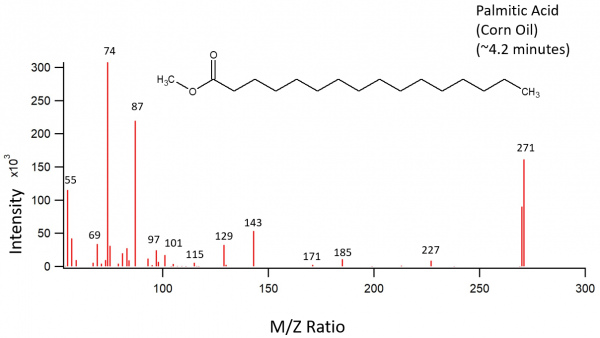
Fatty Acid Methyl Esters
Introduction
The fatty acid composition of a sample can reveal the origins of said sample. Different combinations of fatty acids are associated with different types of oils. This information can be especially useful in the field of archaeology. Dried clay holds lipid residue fairly well, so if a clay pot is crushed up and prepared for GC-MS analysis, the fatty acid composition of the oils in the clay can be discovered. This process can also be carried out with modern cooking oils to determine the different fatty acid composition of different oils or even of different brands of oil. This project replicated a procedure of detecting fatty acid methyl esters in oil samples using GC-MS analysis.
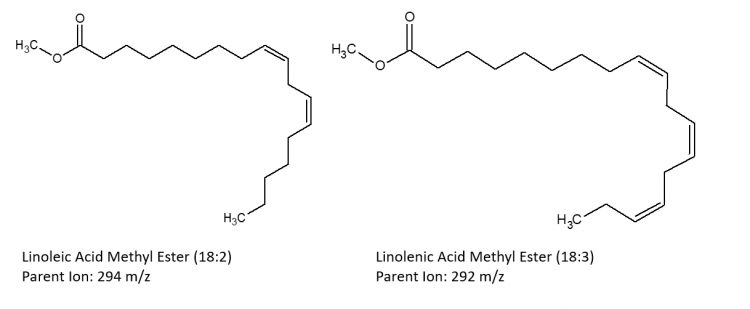
The following images depict the chemical structures of the fatty acid methyl esters that have been detected in the samples. These images were created using ChemSketch.
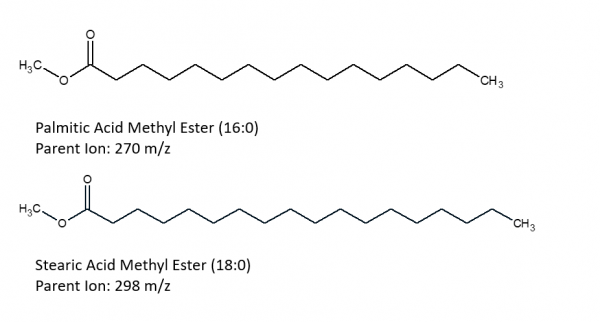
|
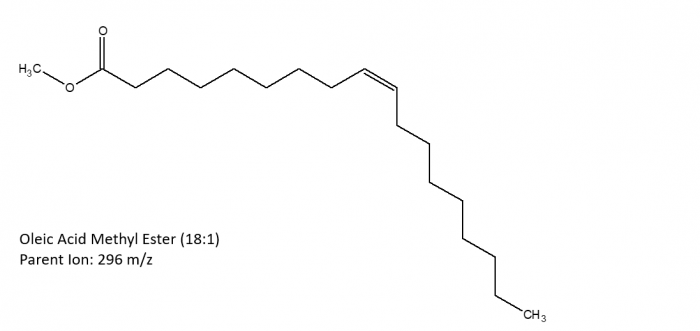
|
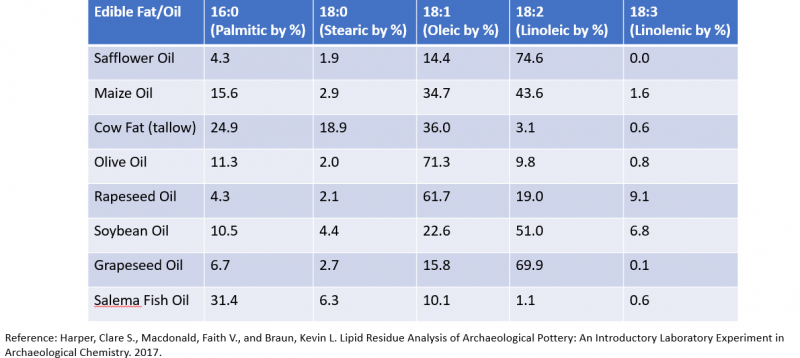
Fatty acid content can be used to help identify the origins of different types of oils. Each type of oil has a unique amount of different fatty acids. Unsaturated fatty acids are more common in plant materials, while saturated fatty acids are more common in animal fats. Certain plant materials are known to be used for tasks such as dyeing fabrics, so being able to tell what type of plant each pot was used for can determine the purpose of each pot. These values can help archaeologists determine what kind of material a pot was used for if the pot is crushed up and sent in for GC-MS analysis. This is just one of the many applications this fatty acid methyl ester GC-MS analysis technique has in a variety of disciplines. This table shows some experimental values of fatty acid content in different types of oils.
Methods and Materials
Materials
Samples
All samples were obtained from Monmouth College's nutrition laboratory. The grapeseed oil was the brand Carlini. The sunflower oil was virgin sunflower oil of the brand Authentic Menu. The corn oil was "pure" corn oil of the brand Essential Everyday.
Chemicals
Methanol, KOH, pentane, and acetic acid were all purchased from Millipore Sigma.
Sample Preparation/Trasesterifcation
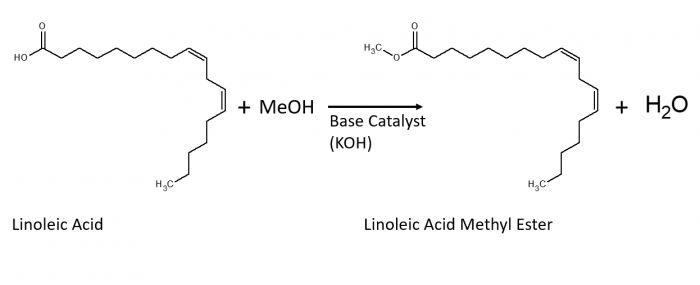
Before an oil sample can be detected by GC-MS analysis, the fatty acids must be methylated to increase their volatility. The process by which this is done is called transesterification. The methyl from the methanol group is added to the carboxylic acid of the fatty acid to create a fatty acid methyl ester.
200 ul of each sample was combined with 40 ul of anhydrous methanol into a microcentrifuge tube. Then 4.3 ul of 5 M KOH was added to the top methanol layer to act as a catalyst for the reaction. The microcentrifuge tubes were then sonicated in a Fisher Scientific model FS30H ultrasonic bath for 30 minutes. After being removed from the bath, 200 ul of 0.1 M acetic acid was added to each microcentrifuge tube to neutralize the base catalyst. Then each tube was centrifuged at 6,000 rpm for 5 minutes in a Capp Denmark model CR-68X tabletop microcentrifuge.
Preparation of sample for GC analysis
After centrifugation, 40 ul of the top layer of each solution was removed and added to a GC vial. Each vial was then diluted with 40 ul of pentane for GC-MS analysis.
Results
GC-MS Analysis
GC-MS Method for the Analysis of Fatty Acid Methyl Esters:
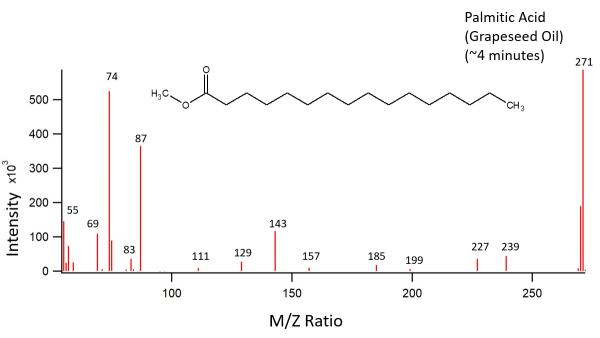
Grapeseed Oil Data
Grapeseed oil was treated as described above. The transesterifcation products were palmitic acid, linoleic acid, and oleic acid. The oleic acid peak was much smaller than expected. While we have no definitive answer as to why this peak was so small, one theory is that the grapeseed oil was old and had undergone oxidation reactions.

|

|

|
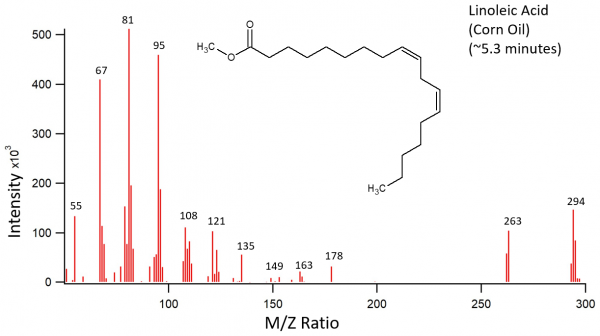
Corn Oil Data
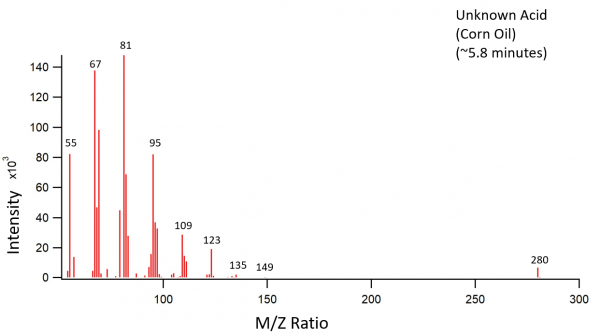
Corn oil was treated as described above. The transesterifcation products were palmitic acid, linoleic acid, and an unknown acid. The third peak on the chromatogram should have been an oleic acid peak according to previously recorded fatty acid contents of corn oil. Instead, the third peak is an unknown fatty acid with a fatty acid methyl ester parent ion of 280 m/z. The chromatogram peaks of the corn oil were also much noisier than the peaks in the other chromatograms.
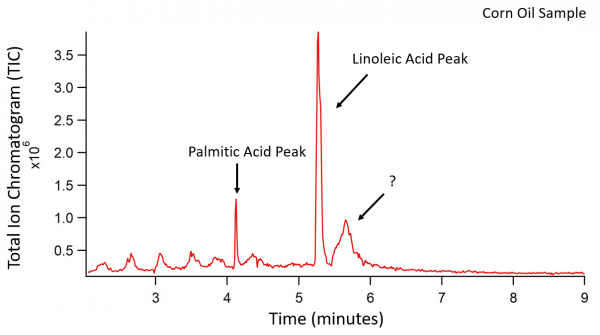
|
|
|
Sunflower Oil Data
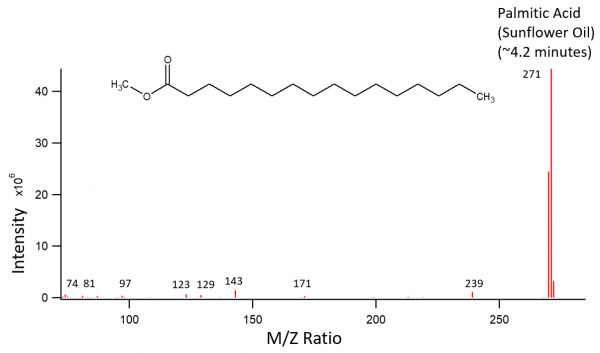
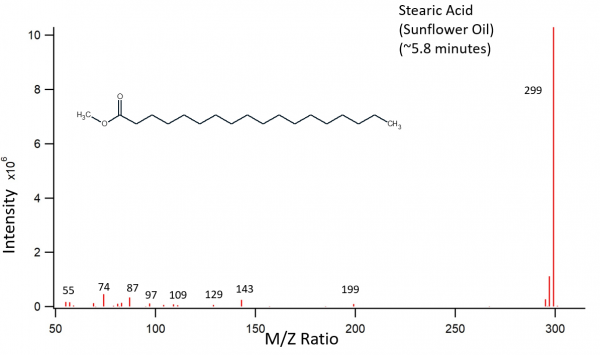
Sunflower oil was treated as described above. The transesterifcation products were palmitic acid, oleic acid, and stearic acid. The largest peak on the chromatogram should have been a linoleic acid peak according to previously recorded fatty acid contents of sunflower oil. Instead, the largest peak is oleic acid, which should have had a much lower content. The third peak is also much smaller than previous data would suggest, and is stearic acid. The stearic acid peak should be very small according to previous data. The largest peak appears to be split as well as wider than one would hope for a GC-MS chromatogram. This could be due to a possible overloading of the column with this particular sample.

|
|

|
|
Discussion
The results of this experiment show that this technique for analyzing fatty acid methyl-esters in oil samples will produce similar to expected values. The issues with some of the peaks representing different acids could be due to oxidation that happened within the oils because of the age of the oils. The corn oil chromatogram peaks were very noisy compared to the other chromatograms. A repeat of this experiment with new oils could eliminate the possibility of this causing issues in the experiment. Another recommendation for this experiment would be to use a slightly larger volume of samples for GC-MS analysis to prevent loss of a great amount of sample due to evaporation.