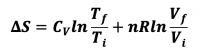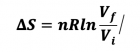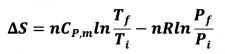PChem312 f20 w13
Monday, Nov 9, 2020
We will now highlight how to calculate entropy (ΔS) for four processes involving ideal gases:
- 1) Reversible Isothermal Expansion or Compression
- ViTi --> VfTi
- 2) Constant P, V temperature change
- ViTi --> ViTf
- PiTi --> PiTf
- 3) Changes in both V and T, constant P
- ViTi -->V fTf
- 4) Changes in both P and T, constant V
- ViTi -->V fTf
Example Problem 1
- (Please write out your complete solution and email to bes by next class period)
Consider a gas undergoing the following change under constant pressure (1.00 bar) conditions; calculate the change in entropy (ΔS): Vi = 10.0 L; Ti = 298 K; Tf = 398 K
Example Problem 2
- (Please write out your complete solution and email to bes by next class period)
Consider a gas undergoing the following change under constant volume (10.0 L) conditions; calculate the change in entropy (ΔS): Pi = 1.00 bar; Ti = 298 K; Tf = 398 K
Wednesday, Nov 11, 2020
- Review contributions to ΔS due to ΔT, ΔV, ΔP for ideal gas/closed system.
- Entropy of Phase Changes...
...continuing numbering from above...
- 5) Solid --> Liquid phase change (fusion/melting/freezing)
- - under constant pressure
- - under constant volume
- 6) Liquid --> gas phase change (vaporization/boiling/condensation)
- - under constant pressure
- - under constant volume
Example Problem 3
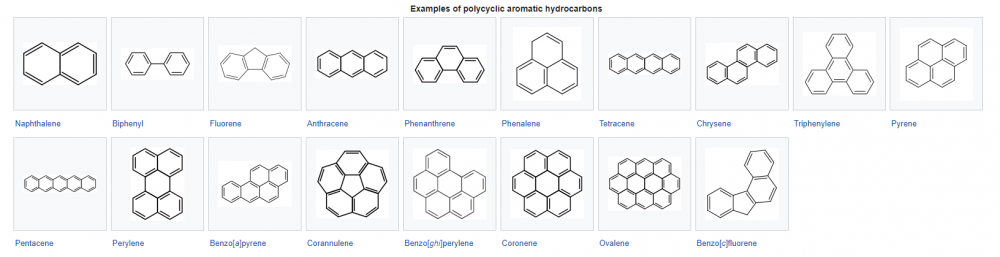
(Please write out your complete solution and email to bes by next class period) Poly aromatic hydrocarbons (<-- wiki link here) represent and interesting set of compounds.
Calculate the entropy of fusion for 5 PAHs.
Example Problem 4
(Please write out your complete solution and email to bes by next class period) Calculate the entropy of vaporization for methanol, ethanol, and propanol under both constant pressure and constant volume conditions.