WebMO Electrostatic Potential maps
(4/23/20, bes)
Exercise 5: Electrostatic Potential Maps
Electrostatic potential maps show the electron distribution (ie. the composite wavefunction for all electrons) within a molecular frame. Did you get that ? <--these pictures are a visual representation of the composite electron wavefunctions.
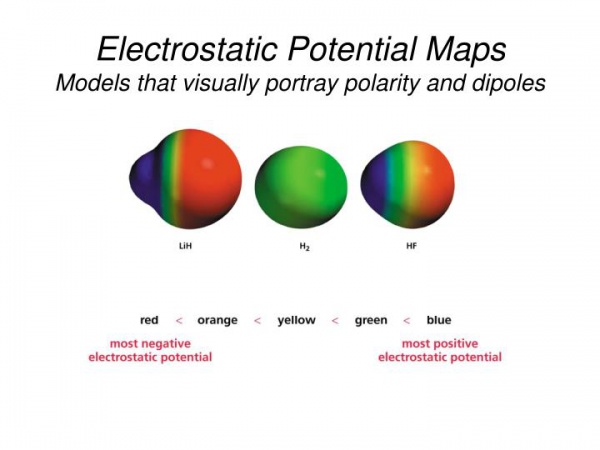
The figure below shows an example of the electrostatic potential maps for LiH, H2, and HF.
- H2, the electrons are evenly distributed among the two H-atoms (green = neutral),
- LiH, being an ionic compound, the 1s valence electron is transferred to the H-atom showing a Li+/positive charge (blue - on left) and an H-/negative charge (red - on right),
- HF, being a covalent bond with the highly electronegative F-atom showing showing a H+/positive charge (blue - on left) and F-/negative charge (red - on right).
Note: these maps are "surface" maps...electrostatic potential surface maps...and represent what the electron distribution looks like from a external perspective.
Using WebMO/Gaussian please complete the following table by optimizing the geometry based the stated level of theory and basis set:
Activity 1: Generating an Electrostatic Potential Map for Benzene (C6H6)
Note: this calculation is a 2-step process:
- 1) Optimize Geometry, then,
- 2) Calculate Molecular Orbitals.
Step 1: Optimize Geometry for Benzene
- - Click “New Job”, then “Create New Job”. The Build Molecule window opens,
- - Build C6H6 using the "fragments:
- ... from the menu bar, select Build: Fragments, then
- - Category: --> Rings
- - Fragment: --> Benzene
- - OK
- ...then a single click in the drawing area will generate a benzene ring.
- - Choose Clean-Up > Comprehensive – Mechanics,
- - Symmetrize the molecule...(click "red" D6h* <-- 10th item down in left side menu),
- - Click the blue “continue” arrow in the lower right side of the Build Molecule window.
- - Setup the job as
- - Name: C6H6 (should be this already)
- - Calculation: Geometry Optimization
- - Theory: Hartree-Fock
- - Basic Set: Routine: 6-31(G)d
- - Charge: 1
- - Multiplicity: Singlet
- - Click the blue “continue” arrow to submit the job.
Step 2: Calculate Molecular Orbitals for Benzene
- - Once the job is complete, click on the hyperlinked name, C6H6 to open the “View Job” window.
- - Click the "New Job Using This Geometry" button at the bottom of the window, then click the blue “continue” arrow,
- - Setup the job:
- - Name: C6H6 (should be this already)
- - Calculation: Molecular Geometry
- - Theory: Hartree-Fock
- - Basic Set: Routine: 6-31(G)d
- - Charge: 1
- - Multiplicity: Singlet
- - Click the blue “continue” arrow to submit the job.
- - Once the job is complete, click on the "molecular orbital" hyperlinked name to view the “View Job” window.
- - Scroll down to the Molecular Orbitals table and look at the bottom:

...and click the magnifying glass next to the "Potential Energy"
Using the energy values and occupancy as your judge, identify the HOMO and LUMO. View these orbitals by click on the magnifying glass icon. The orbitals can be rotated by clicking and dragging in the viewer window.