Difference between revisions of "Acetaminophen Manuscript"
| Line 29: | Line 29: | ||
==Experimental Procedures== | ==Experimental Procedures== | ||
:Procedures for experimental methods should be described in sufficient detail to enable other investigators to repeat the experiments. Names of product manufacturers (with city, state address, catalog number) should be included if alternate sources are deemed unsatisfactory or if the product is of limited availability. Novel experimental procedures should be described in detail, but previously published procedures should be referred to by literature citation of the original detailed explanation, and should include description of any modifications. | :Procedures for experimental methods should be described in sufficient detail to enable other investigators to repeat the experiments. Names of product manufacturers (with city, state address, catalog number) should be included if alternate sources are deemed unsatisfactory or if the product is of limited availability. Novel experimental procedures should be described in detail, but previously published procedures should be referred to by literature citation of the original detailed explanation, and should include description of any modifications. | ||
| + | |||
:Acetaminophen, H2O2 (30%), dimethyl sulfoxide, and Horseradish peroxidase (Type ??) were purchased from Sigma Aldrich. All other reagents and solvents were of the highest grade available…. Dioxane, buffers, methanol, dichloromethane | :Acetaminophen, H2O2 (30%), dimethyl sulfoxide, and Horseradish peroxidase (Type ??) were purchased from Sigma Aldrich. All other reagents and solvents were of the highest grade available…. Dioxane, buffers, methanol, dichloromethane | ||
| + | |||
''Acetaminophen Oxidation'' | ''Acetaminophen Oxidation'' | ||
:Acetaminophen oxidations were carried out with 50mM acetaminophen, 0-50mM H2O2, 0-9.7nm HRP, in 25% dioxane to aqueous buffer (pH 9.2). Samples were equilibrated at room temperature and then initiated by the addition of H2O2. Reactions were allowed to go until completion. Acetaminophen oxidation products were then analyzed and isolated. | :Acetaminophen oxidations were carried out with 50mM acetaminophen, 0-50mM H2O2, 0-9.7nm HRP, in 25% dioxane to aqueous buffer (pH 9.2). Samples were equilibrated at room temperature and then initiated by the addition of H2O2. Reactions were allowed to go until completion. Acetaminophen oxidation products were then analyzed and isolated. | ||
| + | |||
''Liquid Chromatography'' | ''Liquid Chromatography'' | ||
:The analytical HPLC system consisted of reversed-phase column and a normal phase column. A binary solvent system with a flow rate of 1.0 mL/minute containing Solvent A, 0.1% trifluoroacetic acid, and Solvent B, 100% acetonitrile, allowed for the separation and quantification of acetaminophen metabolites following oxidation. For reverse phase HPLC, Solvent A was held at 100% for the first minute of the run, followed by a linear transition to 100% solvent B until minute 15 at which Solvent B was held constant for 3 more minutes. For normal phase HPLC, Solvent B was held at 100% for the first minute of the run, followed by a linear transition to 50% solvent A until minute 15 at which Solvent A was held constant for 3. BOth methods gave a total run time of 18 minutes. | :The analytical HPLC system consisted of reversed-phase column and a normal phase column. A binary solvent system with a flow rate of 1.0 mL/minute containing Solvent A, 0.1% trifluoroacetic acid, and Solvent B, 100% acetonitrile, allowed for the separation and quantification of acetaminophen metabolites following oxidation. For reverse phase HPLC, Solvent A was held at 100% for the first minute of the run, followed by a linear transition to 100% solvent B until minute 15 at which Solvent B was held constant for 3 more minutes. For normal phase HPLC, Solvent B was held at 100% for the first minute of the run, followed by a linear transition to 50% solvent A until minute 15 at which Solvent A was held constant for 3. BOth methods gave a total run time of 18 minutes. | ||
| + | |||
The automated flash chromatography system used to purify Acetaminophen metabolites for NMR and MS analysis consisted of … Purification was carried out via a 5.5mg C18 column from () with a flow rate of 18 mL/minute. Solvent A was held constant at 100% for 1 column volume, followed by a linear transition to 75% Acetonitrile over the course of 29 column volumes. Acetaminophen metabolite fractions were collected and the solvent mixtures were removed using a rotary evaporator. | The automated flash chromatography system used to purify Acetaminophen metabolites for NMR and MS analysis consisted of … Purification was carried out via a 5.5mg C18 column from () with a flow rate of 18 mL/minute. Solvent A was held constant at 100% for 1 column volume, followed by a linear transition to 75% Acetonitrile over the course of 29 column volumes. Acetaminophen metabolite fractions were collected and the solvent mixtures were removed using a rotary evaporator. | ||
| + | |||
''Quadruple Time-of-Flight Mass Spectrometry'' | ''Quadruple Time-of-Flight Mass Spectrometry'' | ||
:Electrospray ionization was utilized with a quadruple time-of-flight functioning as a mass analyzer for the analysis of acetaminophen metabolites solubilized in methanol. Samples were introduced to the mass spectrometer using a direct inject UPLC system… WESTERN | :Electrospray ionization was utilized with a quadruple time-of-flight functioning as a mass analyzer for the analysis of acetaminophen metabolites solubilized in methanol. Samples were introduced to the mass spectrometer using a direct inject UPLC system… WESTERN | ||
| + | |||
''Nuclear Magnetic Resonance Spectroscopy'' | ''Nuclear Magnetic Resonance Spectroscopy'' | ||
:H-NMR spectra were obtained using a 400 MHz Bruker WM-500 NMR spectrometer. Spectra were obtained at room temperature from solutions of samples in dimethyl sulfoxide-d6. KNOX | :H-NMR spectra were obtained using a 400 MHz Bruker WM-500 NMR spectrometer. Spectra were obtained at room temperature from solutions of samples in dimethyl sulfoxide-d6. KNOX | ||
| + | |||
''Electron Spin Resonance'' | ''Electron Spin Resonance'' | ||
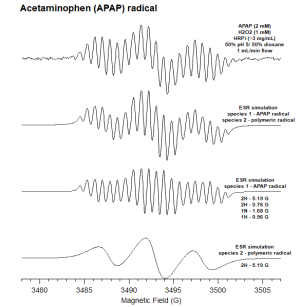
:ESR spectra were measured at room temperature using an ESR spectrometer fitted | :ESR spectra were measured at room temperature using an ESR spectrometer fitted | ||
with a flat cell containing an immobilized enzyme system. The immobilized enzyme system was made using ## HRP bound to aquigel beads. PARAMERTERS Computer simulation of the experimental ESR spectra were carried out using WINSIM. | with a flat cell containing an immobilized enzyme system. The immobilized enzyme system was made using ## HRP bound to aquigel beads. PARAMERTERS Computer simulation of the experimental ESR spectra were carried out using WINSIM. | ||
| + | |||
''Computational Work'' | ''Computational Work'' | ||
Revision as of 18:54, 29 March 2022
This page is being prepared to submit a manuscript for peer review and publication in the Journal of Chemical Research in Toxicology by Matthew Simonson. The goal of this work is to provide foundational evidence for a mechanism of hepatotoxicity caused by radical polymerization of Acetaminophen within the liver as opposed to soley a mechanism of two electron oxidation into NAPQI.
Title Page
- A brief and informative title (preferably fewer than 12 words) will aid in the classification and indexing of the paper. Do not use trade names of drugs, jargon, or abbreviations. Include keywords. List full names and institutional affiliations of all authors, and if differentiation is necessary, indicate the affiliations of each author by the superscript symbols †, ‡, §, ║, ┴, etc. These symbols should also be used to indicate author affiliations different from those stated on the title page and present address information. The author to whom correspondence should be addressed is indicated by an asterisk. It is implicit in listing a person as an author that this individual has agreed to appear as an author of the manuscript.
Table of Contents Graphic
- A Table of Contents (TOC) graphic is published with each manuscript. It is submitted for use in the table of contents and is also used for multiple purposes, including the document abstract and other situations where a representative graphic is required. Create an image that represents the work while adhering to size constraints. Keeping in mind that various devices may be involved, some of the best images are simple, relatively free of text and technical characters, and make use of color for visual impact. It is best to avoid complex structure schemes and small-sized details. The author must submit a graphic in the actual size to be used for the TOC that will fit in an area 8.47 cm by 4.76 cm (3.33 in. by 1.88 in.). Larger images will be reduced to fit within those dimensions. Type size of labels, formulas, or numbers within the graphic must be legible at the specified size. Tables or spectra are not acceptable. Place the TOC graphic after the title page and before the abstract page of the manuscript. All elements of the TOC graphic must be (1) entirely original and (2) created by one or more of the authors. Lastly, this graphic should have no legend.
Abstract
- An abstract should be included with all Articles, Communications, Chemical Profiles, Reviews, and Perspectives. For Articles, Communications, and Chemical Profiles, the abstract should briefly (300 word maximum) present, in one paragraph, the problem and experimental approach and state the findings and conclusions. For Reviews and Perspectives, the abstract should introduce the topic, summarize key points, and state the major conclusions. In all cases, the abstract should be self- explanatory and suitable for reproduction without rewriting. Footnotes or undefined abbreviations may not be used. Avoid the use of jargon, but include keywords relevant to the field to improve indexing and discoverability to potential readers. If a reference must be cited, complete publication data must be given.
- Acetaminophen (APAP) is a common analgesic and an active ingredient in many painkillers such as Tylenol and Percocet. Upon overdose, APAP can lead to toxicity in the liver, which accounts for a striking proportion of acute liver failures in the United States annually. Considering the potential for APAP induced hepatotoxicity, our research group has analyzed APAP and its tendency to oxidize into reactive products through both enzymatic and electrochemical methods of in vitro oxidation, which was visualized through HPLC. Characterization of these APAP oxidation products were carried out using electron spin resonance (ESR), quadruple time-of-flight mass spectrometry (qTOF-MS), 400 MHz proton nuclear magnetic resonance (H-NMR), and cyclic voltammetry (CV). Our findings provide structural insight into potentially deleterious APAP oxidation metabolites formed in the liver during times of APAP overdose formed via a mechanism of radical polymerization.
Introduction
- The introduction should state the purpose of the investigation and its relation to other work in the field. Background material should be brief and relevant to the research described. Detailed or lengthy reviews of the literature should be avoided.
IUPAC NAMES for Acetaminophen (APAP), paracetamol, and intermediates
Introduce APAP and overarching problem
- Acetaminophen (Paracetamol, 4'-Hydroxyacetanilide, 4-Acetamidophenol, or APAP) is one of the most widely used analgesics and antipyretics and can be purchased over the counter. Moreover, APAP overdose remains to be a large problem in the United States and is responsible for a majority of acute liver failures annually (Lee at al; Bernal et al). The difference between a safe dose and a toxic dose is narrow. So it is deceptively easy to take too much and cause a trip to the emergency room, permanent liver damage, or even death.
Introduce NAPQI and Toxicity
- While APAP itself is harmless, APAP metabolic pathways have been investigated by numerous studies which allude to reactive metabolites of APAP being responsible for liver necrosis. The formation of reactive metabolites coupled with glutathione depletion and alkylation of mitochondrial proteins are suggested to be critical initiating events for APAP induced hepatotoxicity (???). Reactive metabolites of APAP form via oxidation of APAP in the liver, which occurs after other nontoxic metabolic pathways such as glucuronidation and sulfation have been saturated (???). The commonly accepted mechanism of oxidative metabolite formation during APAP overdose revolves around the two electron oxidation of APAP into an electrophilic metabolite, N-acetyl-p-benzoquinone imine (NAPQI), via Cytochrome P450 enzymes (CYPs) (Gillette et al. 1981 Dahlin et al. 1984 Nelson et al 1990). NAPQI is then purported to be reduced and detoxified via glutahione (GSH), leading to cysteine and mercapturic acid metabolites to be formed (???). However, once intracellular glutathione is used up, toxicity is suggested to disseminate through NAPQI conjugation with thiol groups of proteins and down stream signaling (???). The formation of NAPQI was determined to be the main deleterious metabolite of APAP oxidation in the liver through indirect measurement of its conjugation with glutathione (Dahlin et al.). The uncertainty associated with indirect metabolic identification raises hesitance to NAPQI being the only actor during APAP overdose.
Alternative Metabolism
- Moreover, oxidation reactions mediated by CYPs involving phenols like APAP have been confirmed to occur by both mechanisms of one electron oxidations or two electron oxidations (Potter and Hinson 1987). Oxidizing APAP by one electron instead of two yields a different set of metabolites, which could play bioactive roles responsible for the hepatotoxicity observed following glutathione conjugation and protein alkylation. Both enzymatic and electrochemical methods have been utilized to model CYP metabolism of phenols as opposed to working with liver microsomes. In one such case, the one electron oxidation of APAP was catalyzed using Horseradich peroxidase, which allowed for the detection of a radical intermediate (1984 and Fischer 1985). This radical intermediate following APAP oxidation could also conjugate with protein thiol groups or GSH and propagate down stream damage just as NAPQI is suggested to do (???). Characterization of the APAP oxidation products following one electron oxidation yielded the identification of a set of APAP polymers consistent with a mechanism of radical polymerization, independent of enzymatic mediation (Potter et al. 1984). Further investigation into such metabolites could shed light on the controversial topic of how APAP induced liver necrosis disseminates. Considering that, the purpose of this work is to revisit the oxidation of APAP and its downstream oxidation products in order to illucidate the radical nature of this reaction.
Experimental Procedures
- Procedures for experimental methods should be described in sufficient detail to enable other investigators to repeat the experiments. Names of product manufacturers (with city, state address, catalog number) should be included if alternate sources are deemed unsatisfactory or if the product is of limited availability. Novel experimental procedures should be described in detail, but previously published procedures should be referred to by literature citation of the original detailed explanation, and should include description of any modifications.
- Acetaminophen, H2O2 (30%), dimethyl sulfoxide, and Horseradish peroxidase (Type ??) were purchased from Sigma Aldrich. All other reagents and solvents were of the highest grade available…. Dioxane, buffers, methanol, dichloromethane
Acetaminophen Oxidation
- Acetaminophen oxidations were carried out with 50mM acetaminophen, 0-50mM H2O2, 0-9.7nm HRP, in 25% dioxane to aqueous buffer (pH 9.2). Samples were equilibrated at room temperature and then initiated by the addition of H2O2. Reactions were allowed to go until completion. Acetaminophen oxidation products were then analyzed and isolated.
Liquid Chromatography
- The analytical HPLC system consisted of reversed-phase column and a normal phase column. A binary solvent system with a flow rate of 1.0 mL/minute containing Solvent A, 0.1% trifluoroacetic acid, and Solvent B, 100% acetonitrile, allowed for the separation and quantification of acetaminophen metabolites following oxidation. For reverse phase HPLC, Solvent A was held at 100% for the first minute of the run, followed by a linear transition to 100% solvent B until minute 15 at which Solvent B was held constant for 3 more minutes. For normal phase HPLC, Solvent B was held at 100% for the first minute of the run, followed by a linear transition to 50% solvent A until minute 15 at which Solvent A was held constant for 3. BOth methods gave a total run time of 18 minutes.
The automated flash chromatography system used to purify Acetaminophen metabolites for NMR and MS analysis consisted of … Purification was carried out via a 5.5mg C18 column from () with a flow rate of 18 mL/minute. Solvent A was held constant at 100% for 1 column volume, followed by a linear transition to 75% Acetonitrile over the course of 29 column volumes. Acetaminophen metabolite fractions were collected and the solvent mixtures were removed using a rotary evaporator.
Quadruple Time-of-Flight Mass Spectrometry
- Electrospray ionization was utilized with a quadruple time-of-flight functioning as a mass analyzer for the analysis of acetaminophen metabolites solubilized in methanol. Samples were introduced to the mass spectrometer using a direct inject UPLC system… WESTERN
Nuclear Magnetic Resonance Spectroscopy
- H-NMR spectra were obtained using a 400 MHz Bruker WM-500 NMR spectrometer. Spectra were obtained at room temperature from solutions of samples in dimethyl sulfoxide-d6. KNOX
Electron Spin Resonance
- ESR spectra were measured at room temperature using an ESR spectrometer fitted
with a flat cell containing an immobilized enzyme system. The immobilized enzyme system was made using ## HRP bound to aquigel beads. PARAMERTERS Computer simulation of the experimental ESR spectra were carried out using WINSIM.
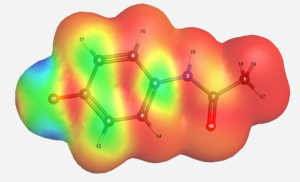
Computational Work
- WebMO was utilized to gather electron spin density information of the acetaminophen radical intermediate. Molecular orbital calculations were gathered using B3LYP/EPR-II as a basis set.
Results
- The results should be presented concisely. Tables and figures should be designed to maximize the presentation and comprehension of the experimental data. The same data should not be presented in more than one figure or in both a figure and a table. Detailed interpretation of results should be reserved for the discussion section of an Article.
Electron Spin Resonance of Radical Acetaminophen Intermediate
Enzymatic Oxidation of Acetaminophen
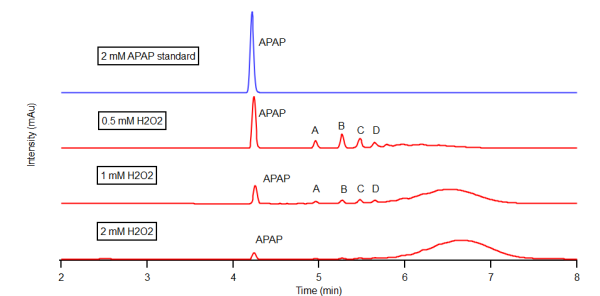
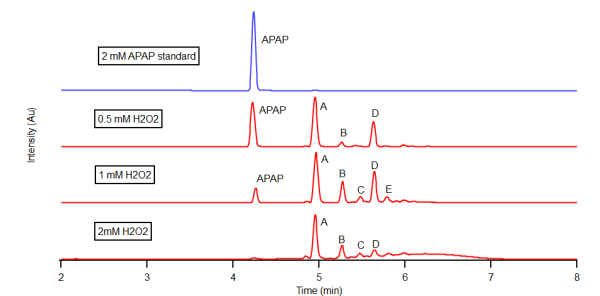
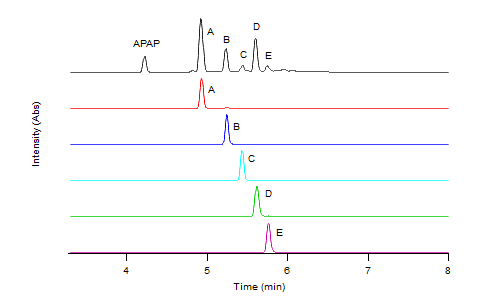
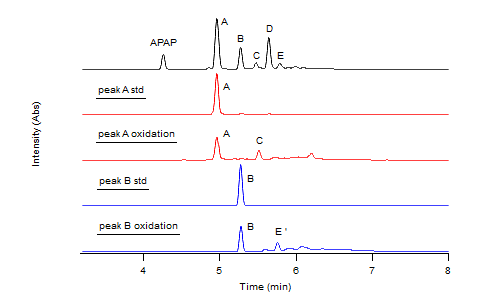
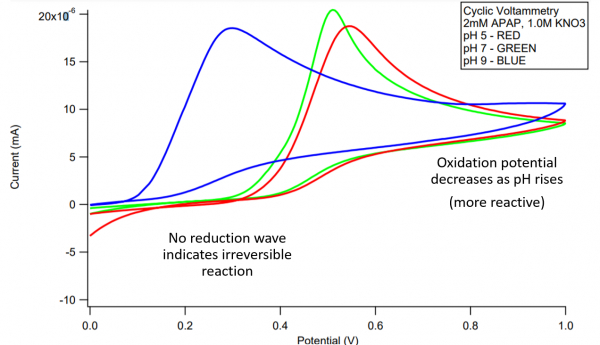
- The enzymatic oxidation of APAP was observed by reverse phase HPLC with a diode array detector set at 270 nm in a 25% dioxane - pH 7 buffer on the left and a 25% dioxane - pH 9 buffer on the right. The chromatogram on top (blue) serves as a 2mM APAP standard prior to reaction. Increasing concentrations of H2O2 were reacted within the APAP/H2O2/HRP system with each red chromatogram down. These concentrations were 0.5mM, 1mM, and 2mM H2O2 respectively. Results indicate the formation of chemically unique products present at both pH 7 and 9, A, B, C, D. However, the formation of these products were maximized in the pH 9 buffer. As H2O2 concentration rose within the pH 7 buffer, increasingly nonpolar products grew in concentration as signified by the hump in the 6-7 minute range.
Flash Purification of APAP Metabolites
Mass Spectrometry of Acetaminophen Metabolites
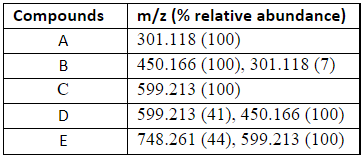
- RERUN MS using HPLC in order to get more peaks
- The data represents a partial set of MS molecular ions as a result of the
- flash chromatography purified oxidation products isolated from the APAP/H2O2/HRP
- oxidation reaction. ESI-MS data are m/z + 1 and contain relative abundance of
- each molecular ion.
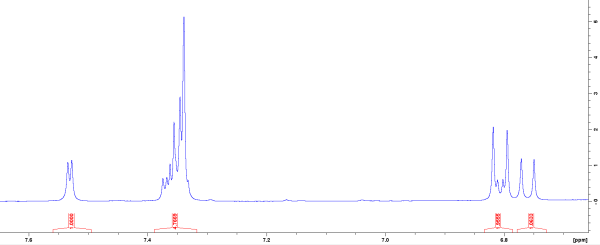
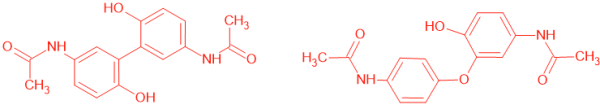
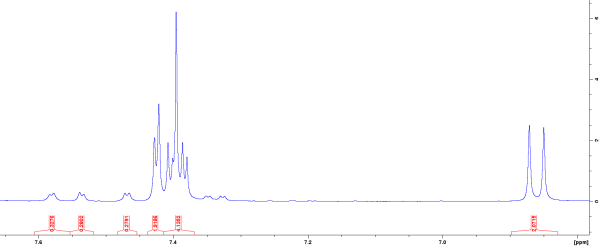
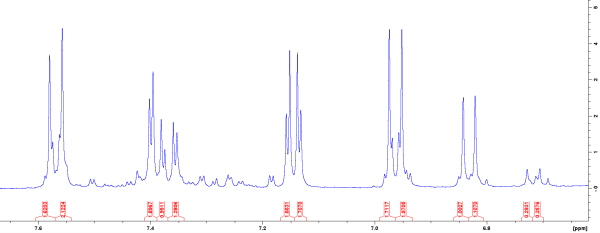
Nuclear Magnetic Resonance of Early Acetaminophen Metabolism
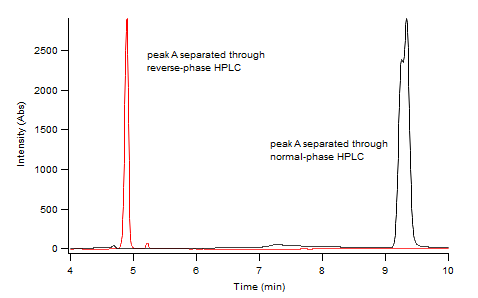
Reverse Phase versus Normal Phase Separation of diAPAP
Early Metabolite Oxidation
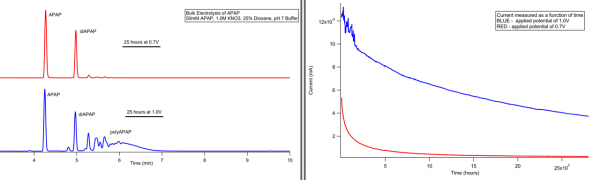
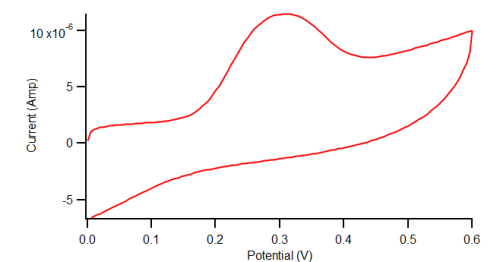
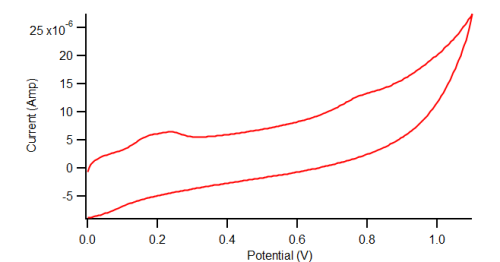
Electrochemical Oxidation of Acetaminophen
Cyclic Voltammetry of diAPAP
Discussion
- The purpose of the discussion is to interpret the results and to relate them to existing knowledge in the field in as clear and brief a fashion as possible. Information given elsewhere in the manuscript should not be repeated in the discussion. Extensive reviews of the literature should be avoided.
Funding Information
- Authors are required to report ALL funding sources and grant/award numbers relevant to this manuscript. Enter all sources of funding for ALL authors relevant to this manuscript in BOTH the Open Funder Registry tool in ACS Paragon Plus and in the manuscript to meet this requirement. See http://pubs.acs.org/page/4authors/funder_options.html for complete instructions.
Acknowledgment
- This section should acknowledge technical assistance, advice from colleagues, gifts, etc. Permission should be sought from persons whose contribution to the work is acknowledged in the manuscript.
Abbreviations
- Abbreviations are used in ACS Journals without periods. Standard abbreviations should be used throughout the manuscript. All nonstandard abbreviations should be kept to a minimum and must be defined in the text following their first use.
Footnotes
- When footnotes are necessary to express some relevant thoughts, these should be included as a parenthetical statement, placed next to the related text to ensure visibility. Footnotes should not be included in the reference list.
Tables
- Tabulation of experimental results is encouraged when this leads to more effective presentation or to more economical use of space. Tables may be created using a word processor’s text mode or table format feature. The table format feature is preferred. Ensure each data entry is in its own cell; no listing of data by using bullets or numbering. If the text mode is used, separate columns with a single tab and use a line feed (return) at the end of each row. Tables should be numbered consecutively with Arabic numerals. Provide a brief title with each table and a brief heading for each column. Clearly indicate the units of measure (preferably SI). Data should be rounded to the nearest significant figure. Explanatory material referring to the whole table is to be included as a footnote to the title (a). Footnotes in tables should be given lower case letter designations and cited in the tables as italicized superscripts. All tables should be cited in the text in consecutive order.
Previously published tables that are being borrowed or adapted from another source require permission from the copyright holder. Once permission is obtained, the permission letter should be uploaded to the submission under the tag “Other Files for Editors Only.” Also, the copyright holder’s preferred credit line should be included in the table’s legend.
Figures and Figure Legends. Line drawings, graphs, stereograms, histograms, and black and white (or color) photographs are all classified as figures and should be numbered consecutively with Arabic numerals in order of citation. Figure legends should be placed after the tables as a single list with the figures following. The figures must be embedded in the same document as the article and not submitted separately as Supporting Information. It may help to print the manuscript on a laser printer to ensure all artwork is clear and legible. See below for guidelines for preparing publication quality illustrations.
- Authors Photos and Biographies. All authors of Reviews and Perspectives (but not other publication formats) should provide a short biography (100 words or less) to be published with the articles.
Headshots (i.e., photos) of the authors may also be provided for publication with the biography. Photos and accompanying biography should be inserted below the reference list in the manuscript. Each photo must be high resolution (at least 300 ppi). No logos may appear in the photo. Before publication, each author must sign (1) a Model Release Form and (2) either a Copyright Transfer Form, which transfers copyright to ACS, or the Nonexclusive Rights Agreement, by which the copyright holder keeps the copyright but grants ACS permission to use the photo. Please choose either the Copyright Transfer Form or the Nonexclusive Rights Agreement; do not sign both forms.