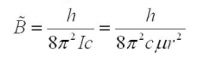Difference between revisions of "HCl Vibrational-Rotational Spectroscopy"
Jump to navigation
Jump to search
| Line 66: | Line 66: | ||
::Reduced mass, ''μ = (m1*m2)/(m1+m2)'' | ::Reduced mass, ''μ = (m1*m2)/(m1+m2)'' | ||
| − | :::: | + | ::::<sup>1</sup>H 1.007825 amu (99.9885%) |
| − | :::: | + | ::::<sup>35</sup>Cl 34.968853 amu (75.78%) |
| − | :::: | + | ::::<sup>37</sup>Cl 36.965903 amu (24.22%) |
::::conversion factor: 1.66054e-27 kg/amu | ::::conversion factor: 1.66054e-27 kg/amu | ||
Revision as of 13:41, 3 April 2020
Adapted from Experiment #38 (Shoemaker, Garland, Nibler, 1989)
See Chapter 8 (Engel), more will be presented in lecture...
Sample Preparation
- a) Pull the HCl (g) off the headspace of a bottle of concentrated HCl using a 60 mL syringe.
- b) deliver the HCl (g) to the gas-sampling IR cell.
IR Gas-Phase Data Collection
- a) Collect data using the highest resolution.
- b) average 32 scans (both background and sample)
- c) Save-As...
- (A complete set of data can be found here.)
Spectral Analysis
- a) load IR data into Igor or Excel.
- b) Use the cursor tool to tabulate the H35Cl and H37Cl data into separate columns,
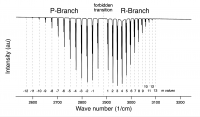
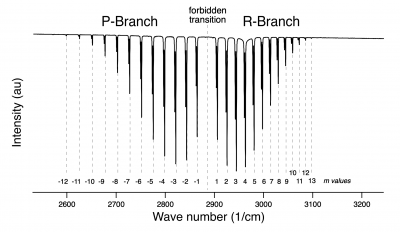
- c) Assign an "m-value" to each transition (note the 'm=0' forbidden transition) with the help of the following graphic (click to make bigger).
Data Analysis
Okay, so you should now have a table, probably in Excel but if you know how to do it in Igor then great (Igor does not offer any advantages here) with heading like this:
| m-value | H35Cl | H37Cl |
| -12 | XXXX | YYYY |
| -11 | XXXX | YYYY |
| -10 | XXXX | YYYY |
| -9 | XXXX | YYYY |
| ... | ... | ... |
| +1 | XXXX | YYYY |
| +12 | XXXX | YYYY |
| +13 | XXXX | YYYY |
- a) Make a plot, m-value verses freq (in 1/cm) for H35Cl data; this will NOT be a straight line but a polynomial curve.
- b) fit a second order polynomial to this data... ie y= ax2 + bx +c, and note the values for a, b, and c in your notes (lab notebook preferred).
- c) During our last lab period you were given a Shoemaker, et al. handout (???) that discussed this lab activity. Eq. 9 was:
ṽ = ṽo + (2Be - 2 αe) m - αe m2, which has the form y = c + bx + ax2
- a = αe
- b = 2Be - 2 αe
- so you can solve for Be...
- d) Now solve for the radius (r), the average internuclear separation of the H35Cl...
- e) repeat above for H37Cl.
Reduced mass calculations
- Reduced mass, μ = (m1*m2)/(m1+m2)
- 1H 1.007825 amu (99.9885%)
- 35Cl 34.968853 amu (75.78%)
- 37Cl 36.965903 amu (24.22%)
- conversion factor: 1.66054e-27 kg/amu
REPORTING
Please send email with the following information:
- H35Cl data
- ṽ0 =
- Be =
- μ (H35Cl) =
- r =
- H37Cl data
- ṽ0 =
- Be =
- μ (H37Cl) =
- r =