Difference between revisions of "ESR Lab Activity"
Jump to navigation
Jump to search
| Line 39: | Line 39: | ||
*Solution A: 0.05 M NaOH; 0.05 grams NaOH (39.997 g/mol) into 25 ml EtOH. | *Solution A: 0.05 M NaOH; 0.05 grams NaOH (39.997 g/mol) into 25 ml EtOH. | ||
*Solution B: 1 M hydroquinone solution. | *Solution B: 1 M hydroquinone solution. | ||
| − | *Procedure: Prepare two | + | *Procedure: Prepare two 60 ml syringes, 1 with solution A and 1 with solution B. |
| + | *Degas the syringe that contains solution B | ||
| + | *Attach to double syringe drive | ||
| + | *Turn on double syringe drive | ||
| + | *Collect data | ||
==Results== | ==Results== | ||
Revision as of 19:58, 21 April 2016
Introduction
We are going to collect an ESR spectrum from a series of substituted hydroquinones.
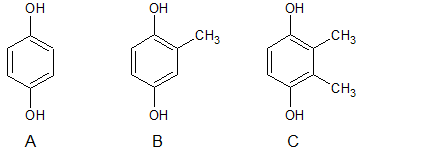
- A=Hydroquinone (110.11 g/mol)
- B=methylhydroquinone (124.14 g/mol)
- C=2,3-dimethylhydroquinone (138.16 g/mol)
| Name | Molar Mass | Sigma Product Number | Cost |
| Hydroquinone | 110.11 g/mol | H9003 | 22.90 / 100g |
| Methylhydroquinone | 124.14 g/mol | 112968 | 59.20 / 250g |
| 2,3-dimethylhydroquinone | 138.16 g/mol | 300756 | 118.00 / 5g |
Experimental
Beaker Method
- Solution A: 1 M NaOH; 1 gram NaOH (39.997 g/mol) into 25 mL EtOH.
- Solution B: 1 M hydroquinone solution.
- Procedure: To 2 ml of 1 M hydroquinone solution add 2-3 drops of solution A. A color change will indicate the reaction has occurred. Quickly transfer colored sample to ESR sample tube, place in ESR spectrometer, tune, and collected data.
Flow Method
- Solution A: 0.05 M NaOH; 0.05 grams NaOH (39.997 g/mol) into 25 ml EtOH.
- Solution B: 1 M hydroquinone solution.
- Procedure: Prepare two 60 ml syringes, 1 with solution A and 1 with solution B.
- Degas the syringe that contains solution B
- Attach to double syringe drive
- Turn on double syringe drive
- Collect data