Difference between revisions of "N-Acetyl L-Tyrosine"
Benstillwell (talk | contribs) |
Benstillwell (talk | contribs) |
||
| Line 13: | Line 13: | ||
[[file:NATw-oDioxane.jpg|500px|thumb|none|Figure 2: 2mM NAT oxidation reacted with 2mM H2O2 and HRP in a beaker 6/5/16]] | [[file:NATw-oDioxane.jpg|500px|thumb|none|Figure 2: 2mM NAT oxidation reacted with 2mM H2O2 and HRP in a beaker 6/5/16]] | ||
| + | [[file:IE Bio Reactor.PNG|500px|thumb|none|Figure 2: 2mM NAT oxidation reacted with 2mM H2O2 and HRP in a beaker 6/5/16]] | ||
[[File:IE69.jpg|500px|thumb|none|Figure 3: 2mM NAT oxidation reacted with 1mM H2O2 using the [[Enzyme Immobilization|Immobilized Enzyme]] technique 6/9/16]] | [[File:IE69.jpg|500px|thumb|none|Figure 3: 2mM NAT oxidation reacted with 1mM H2O2 using the [[Enzyme Immobilization|Immobilized Enzyme]] technique 6/9/16]] | ||
Revision as of 20:15, 11 July 2016
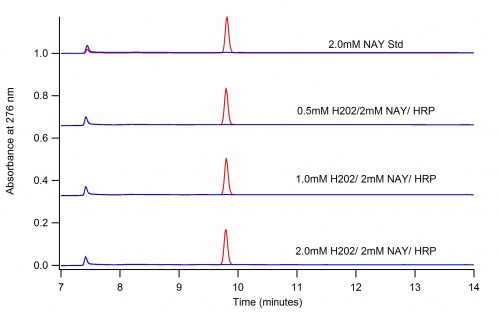
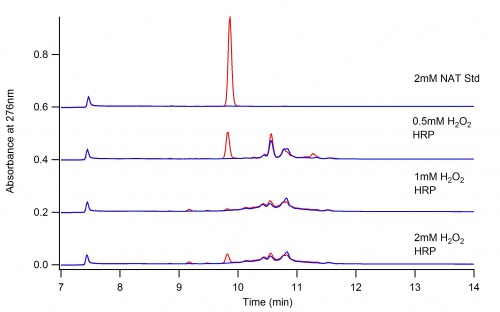
This page depicts the oxidation of N-Acetyl L-Tyrosine (NAT) by Horse Radish Peroxidase (HRP)
The Dioxane Conundrum
The following two graphs depict the difference in reaction when dioxane is present and when it is not. The conclusion is that dioxane quenches the oxidation of NAT.
Beaker Reaction vs. Immobilized Enzyme Bio-Reactor
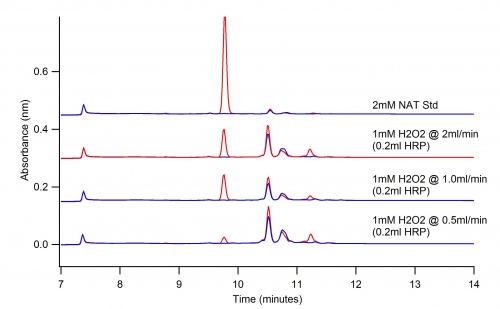
Concentration Test
These graphs depict the difference in concentration of NAT and H2O2. Increasing the concentration will help to obtain more products.
Flash Chromatography
The objective of flash chromatography is to isolate peaks seen in the HPLC. Running flash chromatography allows for the product to be isolated.
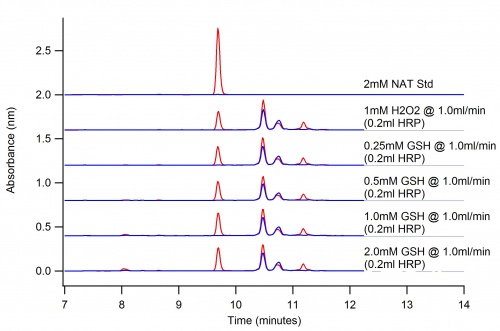
Antioxidants in Reaction
The following graph depicts the oxidation of 2mM NAT by H2O2 with various concentrations of ascorbic acid in pH 5 buffer.
The following is the oxidation of 2mM NAT by H2O2 with various concentrations of reduced GSH in pH 5 buffer.