Difference between revisions of "HPA Reactions"
| Line 23: | Line 23: | ||
[[File:IE HPA7 02.jpg|500px|thumb|Left|IE HRP HPA/H202 (6/20/16)]] | [[File:IE HPA7 02.jpg|500px|thumb|Left|IE HRP HPA/H202 (6/20/16)]] | ||
[[File:IE HPA7 01.jpg|500px|thumb|none|IE HRP HPA/H202 (6/20/16)]] | [[File:IE HPA7 01.jpg|500px|thumb|none|IE HRP HPA/H202 (6/20/16)]] | ||
| + | |||
| + | |||
| + | |||
| + | Decreasing the Concentration to just 20mM brought the resurgence of the peaks of products. These peaks will be separated in flash chromatography given that they are high enough in yield to be detected by the instrument. | ||
| + | [[File:IE HPA9 02.jpg|500px|thumb|Left|IE HRP HPA/H202 (6/23/16)]] | ||
| + | [[File:IE HPA9 01.jpg|500px|thumb|none|IE HRP HPA/H202 (6/23/16)]] | ||
Revision as of 20:13, 23 June 2016
4-Hydroxyphenylacetic acid (HPA)
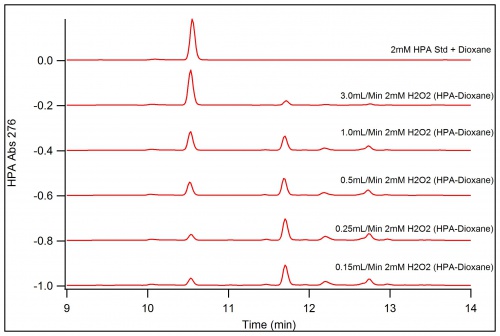
Reaction of 50/50 HPA-Dioxane Solution in Buffer with various H202 concentrations and Horse Radish Peroxidase (HRP)
Using the Immobilized Enzyme technique, a reaction of HPA-Dioxane and H202 with Immobilized HRP
Trying out different flow speeds with IE to try and see different products. With the greater absorption of HPA There is only a slight change in the product of the reactions
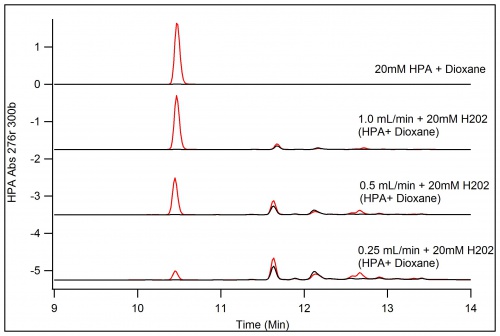
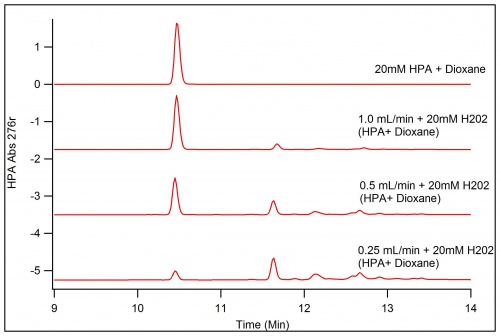
To increase the amount of product extractable, the concentration of HPA was increased to 50mM as well as the H202 concentration, however the same amount of products does not appear as well as the formation of polymers that block products at the 0.25mL/Min run of the immobilized enzyme.
Decreasing the Concentration to just 20mM brought the resurgence of the peaks of products. These peaks will be separated in flash chromatography given that they are high enough in yield to be detected by the instrument.