Difference between revisions of "Acetaminophen Induced Toxicity"
| Line 40: | Line 40: | ||
==Results== | ==Results== | ||
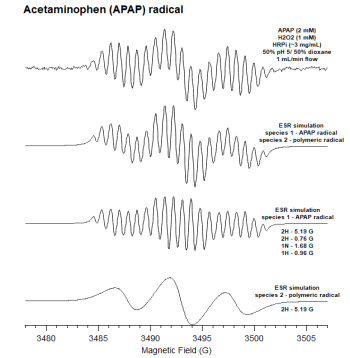
| − | [[File:APAP_ESR_spectra(2).png|left| | + | [[File:APAP_ESR_spectra(2).png|left|350px]]Oxidation of APAP through the IE-ESR system allowed for the detection of a free radical (Figure 1). Low resolution modulation (0.5 G) allowed for a high resolution ESR spectrum with high signal to noise as illustrated in spectrum A. The assignment of hyperfine coupling constants was done using computer software to give a simulation of the full experimental spectrum in spectrum B. Mathematical removal of the signal associated with species 2 yielded the simulated spectrum C while the removal of the species 1 signal resulted in spectrum D. The assignment of the hyperfine coupling values of spectrum B (APAP radical) is summarized in Table # due to the removal of the signal associated with further polymerized APAP radical background noise being mathematically removed. The formation of the radical was dependent upon the presence of APAP, HRP, and H2O2. |
==References== | ==References== | ||
Revision as of 00:53, 8 May 2022
An Alternative Mechanism of Acetaminophen Induced Hepatotoxicity
Matthew W. Simonson, Bradley E. Sturgeon
Monmouth College, Department of Chemistry
Abstract
- Acetaminophen (APAP) is a common analgesic and an active ingredient in many painkillers such as Tylenol and Percocet. Upon overdose, APAP can lead to toxicity in the liver, which accounts for a striking proportion of acute liver failures in the United States annually. Considering the potential for APAP induced hepatotoxicity, our research group has analyzed APAP and its tendency to oxidize into reactive metabolites through enzymatic methods of in vitro oxidation, which was visualized through HPLC pairing with a diode array detector. Characterization of these APAP oxidation products were carried out using electron spin resonance (ESR), electrospray ionization tandem mass spectrometry (ESI-MS/MS), and 400 MHz proton nuclear magnetic resonance (H-NMR). Our findings provide structural and mechanistic insight into the radical propagation of potentially deleterious APAP metabolites formed in the liver during times of APAP overdose.
Introduction
- Acetaminophen (Paracetamol, 4'-Hydroxyacetanilide, 4-Acetamidophenol, or APAP) is one of the most widely used analgesics and antipyretics and can be purchased over the counter. Moreover, APAP overdose remains to be a large problem in the United States and is responsible for a majority of acute liver failures annually (1,2). The quantitative difference between a safe dose and a toxic dose is narrow, and varies by individual based on environmental, genetic, and metabolic factors. Hence, it is deceptively easy to take too much APAP and cause a trip to the emergency room, permanent liver damage, or even death.
- While APAP itself is harmless, APAP metabolic pathways have been investigated by numerous studies which allude to the reactive metabolites of APAP being responsible for liver necrosis. The formation of reactive metabolites coupled with glutathione depletion and alkylation of mitochondrial proteins are suggested to be the critical initiating events for APAP induced hepatotoxicity by way of causing mitochondrial permeability transition (MPT), which is an increase in permeability of the inner membrane that may lead to apoptosis or necrosis of liver cells (3). Reactive metabolites of APAP form via oxidation of APAP in the liver, which occurs after other nontoxic metabolic pathways such as glucuronidation and sulfation have been saturated.
- APAP may undergo a one electron oxidation to yield the APAP phenoxyl radical (N-acetyl-p-benzosemiquinone imine; NAPSQI) or a direct two electron oxidation to produce N-acetyl-p-benzoquinone imine (NAPQI) via Cytochrome P450 enzymes (CYPs) (4). The result leads to hepatotoxicity after intracellular supplies of glutathione have been used up. The commonly accepted mechanism of oxidative metabolite formation during APAP overdose revolves around the direct two electron oxidation of APAP into NAPQI (5, 6). NAPQI is then purported to be reduced and detoxified via glutathione (GSH) conjugation through glutathione transferase, leading to cysteine and mercapturic acid metabolites to be formed (7). The formation of NAPQI has been indirectly identified as a metabolite of APAP oxidation within the liver through its conjugation with glutathione (6). The uncertainty associated with this indirect metabolic identification raises hesitance to NAPQI being the only actor during APAP overdose. Moreover, the oxidation of APAP by one electron to form NAPSQI may be independently reduced by GSH, reforming APAP and forming glutathione disulfide (GSSG) (8). Under scenarios of depleted glutathione reserves, this pathway may yield a different set of metabolites, which could play bioactive roles responsible for the MPT and resulting hepatotoxicity observed following APAP overdose.
- Possible metabolites associated with a one electron oxidation of APAP in the liver have been identified using model systems such as enzymatic or electrochemical methods of oxidation in place of liver microsomes. Following a one electron oxidation by a model enzyme, Horseradish peroxidase (HRP), studies have been done to characterize the APAP radical intermediate in solution through electron spin resonance (ESR) and the potential for this radical to from a different set of metabolites that are not NAPQI (9,10). Characterization of the postulated metabolites allowed for the identification of a set of APAP polymers consistent with a mechanism of radical polymerization (11). Deeper investigation into such metabolites and their radical intermediates could shed light on the controversial topic of how APAP induced liver necrosis disseminates. Considering that, the purpose of this work is to revisit the oxidation of APAP and its downstream oxidation products in order to elucidate a better understanding of the chemistry occurring during this reaction.
Materials and Methods
- Acetaminophen, H2O2 (30%), dioxane, methanol, buffer tablets, deuterated dimethyl sulfoxide, and Horseradish peroxidase (Type II) were purchased from Sigma Aldrich.
Acetaminophen Oxidation
- Acetaminophen oxidations were carried out with 50mM acetaminophen, 0-50mM H2O2, 0-9.7nm HRP, in 25% dioxane to aqueous saline buffer (pH 9.2). Samples were equilibrated at room temperature and then initiated by the addition of H2O2. Reactions were allowed to go until completion. Acetaminophen oxidation products were then analyzed and isolated.
Liquid Chromatography
- The analytical HPLC system that was used for analysis was an Agilent 1100 HPLC paired with a diode array detector (190 nm - 850 nm). Both a reverse phase C18 column and a normal phase amine column were utilized for separation. A binary solvent system with a flow rate of 1.0 mL/minute containing Solvent A, 0.1% trifluoroacetic acid, and Solvent B, 100% acetonitrile, allowed for the separation and quantification of acetaminophen metabolites following oxidation. For reverse phase HPLC, Solvent A was held at 100% for the first minute of the run, followed by a linear transition to 100% solvent B until minute 15 at which Solvent B was held constant for 3 more minutes. For normal phase HPLC, Solvent B was held at 100% for the first minute of the run, followed by a linear transition to 50% solvent A until minute 15 at which Solvent A was held constant for 3. BOth methods gave a total run time of 18 minutes.
- The automated flash chromatography system used to purify Acetaminophen metabolites for characterization was a Teledyne Isco CombiFlash Rf 200i system. Purification was carried out via a 5.5mg C18 gold column with a flow rate of 18 mL/minute. Solvent A was held constant at 100% for 1 column volume, followed by a linear transition to 75% Acetonitrile over the course of 29 column additional column volumes. Acetaminophen metabolite fractions were collected and the solvent mixtures were removed using a rotary evaporator.
Mass Spectrometry
- Electrospray ionization was utilized with a quadruple time-of-flight tandem mass analyzer for the analysis of purified acetaminophen metabolites solubilized in methanol. Samples were introduced to the mass spectrometer using direct injection.
Nuclear Magnetic Resonance Spectroscopy
- H-NMR spectra were obtained using a 400 MHz Bruker NMR spectrometer. Spectra were obtained at room temperature with samples solubilized in deuterated dimethyl sulfoxide.
Electron Spin Resonance
- ESR spectra were measured at room temperature using an Bruker EMX EPR Spectrometer fitted with a flat cell containing an immobilized enzyme system. The immobilized enzyme system was made using 1 mL of Affi-Gel® 10 beads and 10 mg HRP and inserted into the flat cell. Computer simulation of the experimental ESR spectra were carried out using WINSIM.
Computational Work
- WebMO and Gaussian were utilized to gather electron spin density information of the acetaminophen radical intermediate. Molecular orbital calculations were gathered using B3LYP/EPR-II as a basis set.
Results
Oxidation of APAP through the IE-ESR system allowed for the detection of a free radical (Figure 1). Low resolution modulation (0.5 G) allowed for a high resolution ESR spectrum with high signal to noise as illustrated in spectrum A. The assignment of hyperfine coupling constants was done using computer software to give a simulation of the full experimental spectrum in spectrum B. Mathematical removal of the signal associated with species 2 yielded the simulated spectrum C while the removal of the species 1 signal resulted in spectrum D. The assignment of the hyperfine coupling values of spectrum B (APAP radical) is summarized in Table # due to the removal of the signal associated with further polymerized APAP radical background noise being mathematically removed. The formation of the radical was dependent upon the presence of APAP, HRP, and H2O2.
References
1. Lee, W. M. (2013) File:Lee; Drug Induced Liver Failure.pdf. Clin Liver Dis. 17. 575-586.
2. Bernal, W., Lee, W. M., Wendon, J., Larsen, F. S., and Williams, R. (2015) Acute liver failure: A curable disease by 2024?. J. Hepatol. 62. 112-120.
3. Hinson, J. A., Reid, A. B., McCullough, S. S., & James, L. P. (2004). Acetaminophen‐Induced Hepatotoxicity: Role of Metabolic Activation, Reactive Oxygen/Nitrogen Species, and Mitochondrial Permeability Transition. Drug Metabolism Reviews, 36(3-4), 805–822.
4. Potter DW, Hinson JA. Mechanisms of acetaminophen oxidation to N-acetyl-P-benzoquinone imine by horseradish peroxidase and cytochrome P-450. J Biol Chem. 1987;262:966–973.
5. Gillette JR, Nelson SD, Mulder GJ, Jollow DJ, Mitchell JR, Pohl LR, Hinson JA. Formation of chemically reactive metabolites of phenacetin and acetaminophen. Adv Exp Med Biol. 1981;136(Pt B):931–950.
6. Dahlin, D. C., Miwa, G. T., Lu, A. Y., and Nelson, S. D. (1984) N-acetyl-p-benzoquinone imine: a cytochrome P-450-mediated oxidation product of acetaminophen. Proc. Natl. Acad. Sci. U. S. A. 81, 1327-1331.
7. Mitchell JR, Jollow DJ, Potter WZ, Gillette JR, Brodie BB. Acetaminophen-induced hepatic necrosis. IV. Protective role of glutathione. J Pharmacol Exp Ther. 1973b;187:211–217.
8. David Ross, Kajsa Norbeck, and Peter Moldeus. (1985) The Generation and Subsequent Fate of Glutathionyl Radicals in Biological Systems. Journal of Biological Chemistry. Vol 260(28) 15028-15032.
9. West P. R. Harman L. S. Josephy P. D. and Mason R. P.(1984) Acetaminophen: enzymatic formation of a transient phenoxyl free radical. Journal of Biochemical Pharmacology. Vol 33(18) 2933-2936.
10. Volker Fischer, Paul R. West, Laura S. Harman, and Ronald P. Mason. (1985) Free-Radical Metabolites of Acetaminophenand a Dimethylated Derivative. Environmental Health Perspectives Vol 64 127-137.
11. Potter, D. W., Miller, D. W., and Hinson, J. A. (1985) "Identification of Acetaminophen Polymerization Products Catalyzed by Horseradish Peroxidase." J. Biol. Chem. 260. 12,174-12,180.