Difference between revisions of "Acetaminophen Project"
| Line 85: | Line 85: | ||
==References== | ==References== | ||
1.) Hinson, Jack A., Dean W. Roberts, and Laura P. James. "Mechanisms of Acetaminophen-Induced Liver Necrosis." Handbook of Experimental Pharmacology Adverse Drug Reactions (2009): 369-405. Web. | 1.) Hinson, Jack A., Dean W. Roberts, and Laura P. James. "Mechanisms of Acetaminophen-Induced Liver Necrosis." Handbook of Experimental Pharmacology Adverse Drug Reactions (2009): 369-405. Web. | ||
| + | 2.) Potter, David W., Dwight W. Miller, and Jack A. Hinson. "Identification of Acetaminophen Polymerization Products Catalyzed by Horseradish Peroxidase." Journal of Biological Chemistry 260.22 (1985): 12174-2180. Print | ||
Revision as of 14:34, 3 July 2019
This project's goal was to quantify the concentration of Acetaminophen left after a reaction with hydrogen peroxide, Acetaminophen, & horseradish peroxidase. To do this, I made standards with known concentrations of Acetaminophen. Then, we ran the reaction in three separate vials with varying amounts of peroxide with the constant amounts of Acetaminophen and horseradish peroxidase (HRP).
My research partner, Josie Welker, ran a similar experiment with HPA, the link to his page can be found here.
Introduction
Acetaminophen(APAP) is an active ingredient in many over-the-counter and prescription painkillers, such as Tylenol and Oxycodone. APAP is also responsible for approximately 50% of the cases of acute liver-failure in the United States and Great Britain(1). Treatments for acetaminophen-induced liver injury(AILI) are limited. The current mechanism for AILI is the production of hepatotoxic NAPQI as a metabolite in an enzymatic, two-electron oxidation(1). However, evidence also supports a one electron oxidation(2). In on effort to reduce the mortality rate of acetaminophen overdose, we are investigating the identity of single-electron, oxidation products and their bioactivity as an alternative mechanism for AILI. Currently, there is no effective treatment for AILI. The first step towards achieving this would be to definitively determine the mechanism of acetaminophen metabolite toxicity.
Acetaminophen Standards
In order for our samples to be tested, a 2 mM Acetaminophen pH 5 solution, Hydrogen peroxide stock solution, and horseradish peroxidase solution were made.
2 mM Acetaminophen Buffer Stock Solution
Put the 2 mM Acetaminophen standard into a jar, and add the pH 5 buffer tablet. Stir until dissolved.
Hydrogen Peroxide Stock Solution
Add 5 mL of water into a scintillation vial. Add 283 uL of 30% Hydrogen Peroxide to the water. The solution is now at about 0.413 M.
Horseradish Peroxidase Solution
Preparing The Final samples
Measure out 5 mL of Acetaminophen into four different scintillation vials. Add differing amounts of Hydrogen peroxide solution into each of the vials (3uL, 6uL, 12uL, & 24uL). Add 10uL horseradish peroxidase solution to each of the vials.
Instrumentation
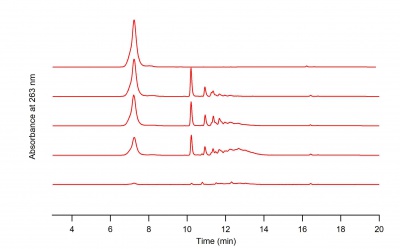
After preparing our solutions, we tested them on the HPLC to see where the peaks were at different concentrations of hydrogen peroxide.
The following link provides insight into how the HPLC works and how a user can correctly use the instrument.
What the HPLC Data Tells us
The Acetaminophen samples were ran at a wavelength of 263 nm because that is the lambda max for acetaminophen. Running the samples at this wavelength shows us how much absorption occurs at that wavelength.
Data Analysis
The data from the HPLC was exported as an arw file in order to be analyzed. This file was then loaded into Igor, which we used to make chromatograms. Chromatograms allowed us to gain a better understanding of the how each concentration of Acetaminophen absorbs at 263 nm for our standards, and how each sample absorbs at 263 nm. We used the information from the chromatograms to make standard curves in excel. The slopes of the standard curves gave us an equation we could use to find the concentration of a sample using the absorbance.
Gepasi
We used the software "Gepasi" to analyze the kinetics of the reaction between horseradish peroxidase (HRP) and hydrogen peroxide. This program simulates how the compounds will react with each other. The following link describes how to use "Gepasi":
Gepasi Graphs
Copasi
A newer, updated version of Gepasi is Copasi. This software is newer and will be more efficient in getting data to the user. A link on how to navigate and use Copasi is listed below.
References
1.) Hinson, Jack A., Dean W. Roberts, and Laura P. James. "Mechanisms of Acetaminophen-Induced Liver Necrosis." Handbook of Experimental Pharmacology Adverse Drug Reactions (2009): 369-405. Web. 2.) Potter, David W., Dwight W. Miller, and Jack A. Hinson. "Identification of Acetaminophen Polymerization Products Catalyzed by Horseradish Peroxidase." Journal of Biological Chemistry 260.22 (1985): 12174-2180. Print