Difference between revisions of "SAHounsve Spring 2018"
| (16 intermediate revisions by the same user not shown) | |||
| Line 17: | Line 17: | ||
===Proposal=== | ===Proposal=== | ||
The purpose of the experiment is to report the observation of radical products of eugenol produced by horseradish peroxidase (HRP) and hydrogen peroxide by using HPLC. | The purpose of the experiment is to report the observation of radical products of eugenol produced by horseradish peroxidase (HRP) and hydrogen peroxide by using HPLC. | ||
| + | |||
| + | ===Introduction=== | ||
| + | Eugenol is a phenolic derivative that can be extracted from spices and herbs like cinnamon, nutmeg, basil, cloves, and some natural oils. Eugenol is a yellow oily liquid with the characteristic fragrance of cloves. The phenol belongs to the most active natural antioxidants found in essential oils. | ||
| + | [[File:eug_structure.png|400px|thumb|left|Figure 1: Chemical structure of eugenol]] | ||
| + | It is well known that natural antioxidants extracted from herbs and spices have vast antioxidant activity and are used in numerous food applications.<sup>1</sup> Most of the antioxidant potential of herbs and spices is due to the redox properties of their phenolic compounds, which permits them to act as reducing agents, hydrogen donors, and singlet oxygen quenchers.<sup>1</sup> Phenols are able to donate hydrogen atoms of phenol hydroxyl groups in reaction with peroxyl radicals that can produce stabilized phenoxyl radicals, thus terminating lipid peroxidation chain reactions.<sup>2</sup> | ||
===Instruments to be used=== | ===Instruments to be used=== | ||
| Line 24: | Line 29: | ||
Eugenol standard for HPLC: | Eugenol standard for HPLC: | ||
15.5 ul of eugenol in 0.1 mM PBS with 10% ethanol, pH 7.4 and 1% tween. Used method "eugenol_SH_020918_b". | 15.5 ul of eugenol in 0.1 mM PBS with 10% ethanol, pH 7.4 and 1% tween. Used method "eugenol_SH_020918_b". | ||
| + | |||
| + | Different proportions of solvents such as 0.1% TFA as eluent B and acetonitrile as eluent A were used for the separation. The multiple gradient used for the chromatographic separation consisted of different proportions of eluent A/B (0:100 for 1-15 mins, 80:20 for 15-25 min, and 0:100 for 25-30 min). The mobile phase flow rte was 1.0 mL/min, the sample injection volume 10 μL, and the chromatograph was monitored at 280 nm. | ||
Oxidation reaction: | Oxidation reaction: | ||
| − | 1. Eugenol stock substrate of | + | |
| − | 2. 0.25 M | + | 1. 1 mM Eugenol stock substrate of pH 7.4 PBS with 10% ethanol |
| + | |||
| + | 2. 0.25 M H<sub>2</sub>O<sub>2</sub> Stock | ||
| + | |||
3. HRP Stock | 3. HRP Stock | ||
| + | {| class="wikitable" | ||
| + | |- | ||
| + | ! Trial !! Eugenol (1 mM) !! H<sub>2</sub>O<sub>2</sub> (0.25 M) !! HRP | ||
| + | |- | ||
| + | | 1 || 5μL || 10μL || 10μL | ||
| + | |- | ||
| + | | 2 || 5μL || 5μL || 10μL | ||
| + | |- | ||
| + | | 3 || 5μL || 2.5μL || 10μL | ||
| + | |- | ||
| + | | 4 || 5μL || 0μL || 10μL | ||
| + | |} | ||
| + | Table 1: Series of mixtures made to oxidize eugenol. Eugenol and HRP had the same amount in each trial, Where H<sub>2</sub>O<sub>2</sub> had varying amounts. | ||
| + | |||
| + | ===Results and Discussion=== | ||
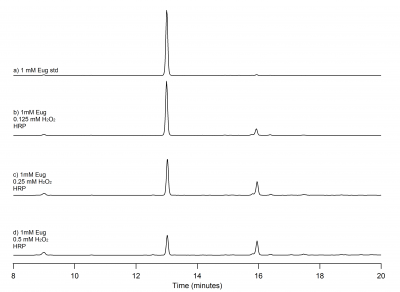
| + | [[File:eug_trails_only_a.png|400px|thumb|left|Figure 2: HPLC chromatograph of 1 mM eugenol standard and oxidation mixtures. The eugenol standard is present at 13 minutes, at 9 minutes there is a small peak that is unknown at what is present here. At 16 minutes, there is a peak that could be a possible radical product.]] | ||
| + | Eugenol oxidized by HRP activated by hydrogen peroxide in solution is seen in Figure 2. | ||
| + | The eugenol peak is at 13 minutes, with the top being the 1 mM eugenol standard only. The following peaks below the eugenol standard are increasing amounts of hydrogen peroxide. As the concentration of hydrogen peroxide increases, the eugenol peak decreases. This means that eugenol is being oxidized. | ||
| + | As eugenol is being oxidized, the peak at 16 minutes could be a possible radical product because this peak increases in height as eugenol decreases. | ||
| + | There is another peaks at 9 minutes, however we do not know what this peak is at this time.There is a peak at 16 minutes that could be a possible radical product. As the concentration of H<sub>2</sub>O<sub>2</sub> increases, the eugenol peak decreases and the possible radical product at 16 minutes increases, indicating that this could a possible radical product. | ||
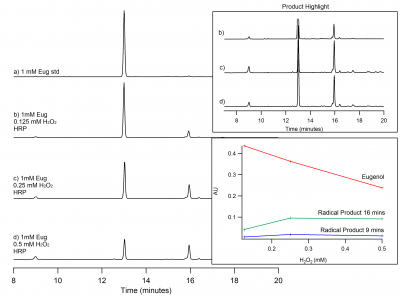
| + | [[File:eug_trial_c.png|400px|thumb|left|Figure 3: HPLC chromatograph with product highlights and kinetic graph. Kinetic graph shows relationship between eugenol and H<sub>2</sub>O<sub>2</sub>.]] | ||
| + | |||
| + | To further investigate if this is a radical product from eugenol is to implement Flash Chromatography and Electron Paramagnetic Resonance (EPR). By utilizing Flash Chromatography, the sample can be injected in the instrument where the mixture is separated into fractions. The specific fractions that contained either the peak at 9 minutes or 16 minutes can be used on either Gas Chromatography –Mass Spectrometry (GC-MS) or Nuclear Magnetic Resonance (NMR) to determine the structure of the radical product. Once the structure of the product is determined, we need to know how this product is related to eugenol. To do so, we would use EPR to determine if it is a free radical and where the unpaired electron is on the product’s structure. | ||
| − | |||
| − | |||
| − | |||
===References=== | ===References=== | ||
| + | |||
| + | 1. Hakkim, Lukmanul F., C. Gowri Shankar, and S. Girija. Chemical Composition and Antioxidant Property of Holy Basil (Ocimum sanctum L.) Leaves, Stems, and Inflorescence and Their in Vitro Callus Cultures. J. Agric. Food Chem., 2007, 55, 9109-9117. | ||
| + | |||
| + | 2. Mastelić, Josip, et al. Comparative Study on the Antioxidant and Biological Activities of Carvacrol, Thymol, and Eugenol Derivatives. J. Agric. Food Chem., 2008, 56, 3989-3996. | ||
| + | |||
| + | 3. Sipe, H. J., Lardinois, O. M., & Mason, R. P. Free Radical Metabolism of Methyleugenol and Related Compounds. Chemical Research in Toxicology, 2014, 27(4), 483–489. | ||
| + | |||
===Research pledge=== | ===Research pledge=== | ||
I, Selene Hounsve, have read the Chem/Bioc 430 course syllabus and understand the general structure and expectations of the research program. The above material was prepared after consultation, and in conjunction with my research advisor. | I, Selene Hounsve, have read the Chem/Bioc 430 course syllabus and understand the general structure and expectations of the research program. The above material was prepared after consultation, and in conjunction with my research advisor. | ||
Latest revision as of 00:08, 11 May 2018
Chemistry/Biochemistry Research 430
Spring 2018
Selene Hounsve
Senior Biochemistry Major
Research Times
Wed: 11AM-2PM
Fri: 4-5 PM
Proposed Research Project
Project Title
Enzymatic Metabolism of Eugenol
General Information
Advisor: Dr. Bradley E. Sturgeon
Proposal
The purpose of the experiment is to report the observation of radical products of eugenol produced by horseradish peroxidase (HRP) and hydrogen peroxide by using HPLC.
Introduction
Eugenol is a phenolic derivative that can be extracted from spices and herbs like cinnamon, nutmeg, basil, cloves, and some natural oils. Eugenol is a yellow oily liquid with the characteristic fragrance of cloves. The phenol belongs to the most active natural antioxidants found in essential oils.
It is well known that natural antioxidants extracted from herbs and spices have vast antioxidant activity and are used in numerous food applications.1 Most of the antioxidant potential of herbs and spices is due to the redox properties of their phenolic compounds, which permits them to act as reducing agents, hydrogen donors, and singlet oxygen quenchers.1 Phenols are able to donate hydrogen atoms of phenol hydroxyl groups in reaction with peroxyl radicals that can produce stabilized phenoxyl radicals, thus terminating lipid peroxidation chain reactions.2
Instruments to be used
HPLC and UV-Vis
Experimental
Eugenol standard for HPLC: 15.5 ul of eugenol in 0.1 mM PBS with 10% ethanol, pH 7.4 and 1% tween. Used method "eugenol_SH_020918_b".
Different proportions of solvents such as 0.1% TFA as eluent B and acetonitrile as eluent A were used for the separation. The multiple gradient used for the chromatographic separation consisted of different proportions of eluent A/B (0:100 for 1-15 mins, 80:20 for 15-25 min, and 0:100 for 25-30 min). The mobile phase flow rte was 1.0 mL/min, the sample injection volume 10 μL, and the chromatograph was monitored at 280 nm.
Oxidation reaction:
1. 1 mM Eugenol stock substrate of pH 7.4 PBS with 10% ethanol
2. 0.25 M H2O2 Stock
3. HRP Stock
| Trial | Eugenol (1 mM) | H2O2 (0.25 M) | HRP |
|---|---|---|---|
| 1 | 5μL | 10μL | 10μL |
| 2 | 5μL | 5μL | 10μL |
| 3 | 5μL | 2.5μL | 10μL |
| 4 | 5μL | 0μL | 10μL |
Table 1: Series of mixtures made to oxidize eugenol. Eugenol and HRP had the same amount in each trial, Where H2O2 had varying amounts.
Results and Discussion
Eugenol oxidized by HRP activated by hydrogen peroxide in solution is seen in Figure 2. The eugenol peak is at 13 minutes, with the top being the 1 mM eugenol standard only. The following peaks below the eugenol standard are increasing amounts of hydrogen peroxide. As the concentration of hydrogen peroxide increases, the eugenol peak decreases. This means that eugenol is being oxidized. As eugenol is being oxidized, the peak at 16 minutes could be a possible radical product because this peak increases in height as eugenol decreases. There is another peaks at 9 minutes, however we do not know what this peak is at this time.There is a peak at 16 minutes that could be a possible radical product. As the concentration of H2O2 increases, the eugenol peak decreases and the possible radical product at 16 minutes increases, indicating that this could a possible radical product.
To further investigate if this is a radical product from eugenol is to implement Flash Chromatography and Electron Paramagnetic Resonance (EPR). By utilizing Flash Chromatography, the sample can be injected in the instrument where the mixture is separated into fractions. The specific fractions that contained either the peak at 9 minutes or 16 minutes can be used on either Gas Chromatography –Mass Spectrometry (GC-MS) or Nuclear Magnetic Resonance (NMR) to determine the structure of the radical product. Once the structure of the product is determined, we need to know how this product is related to eugenol. To do so, we would use EPR to determine if it is a free radical and where the unpaired electron is on the product’s structure.
References
1. Hakkim, Lukmanul F., C. Gowri Shankar, and S. Girija. Chemical Composition and Antioxidant Property of Holy Basil (Ocimum sanctum L.) Leaves, Stems, and Inflorescence and Their in Vitro Callus Cultures. J. Agric. Food Chem., 2007, 55, 9109-9117.
2. Mastelić, Josip, et al. Comparative Study on the Antioxidant and Biological Activities of Carvacrol, Thymol, and Eugenol Derivatives. J. Agric. Food Chem., 2008, 56, 3989-3996.
3. Sipe, H. J., Lardinois, O. M., & Mason, R. P. Free Radical Metabolism of Methyleugenol and Related Compounds. Chemical Research in Toxicology, 2014, 27(4), 483–489.
Research pledge
I, Selene Hounsve, have read the Chem/Bioc 430 course syllabus and understand the general structure and expectations of the research program. The above material was prepared after consultation, and in conjunction with my research advisor.