Difference between revisions of "Emission Spectra of Different Metals"
Jump to navigation
Jump to search
BroddieD16 (talk | contribs) |
BroddieD16 (talk | contribs) |
||
| (One intermediate revision by one other user not shown) | |||
| Line 1: | Line 1: | ||
| + | [[Light_lab_Pchem|Return to Light lab]] | ||
| + | |||
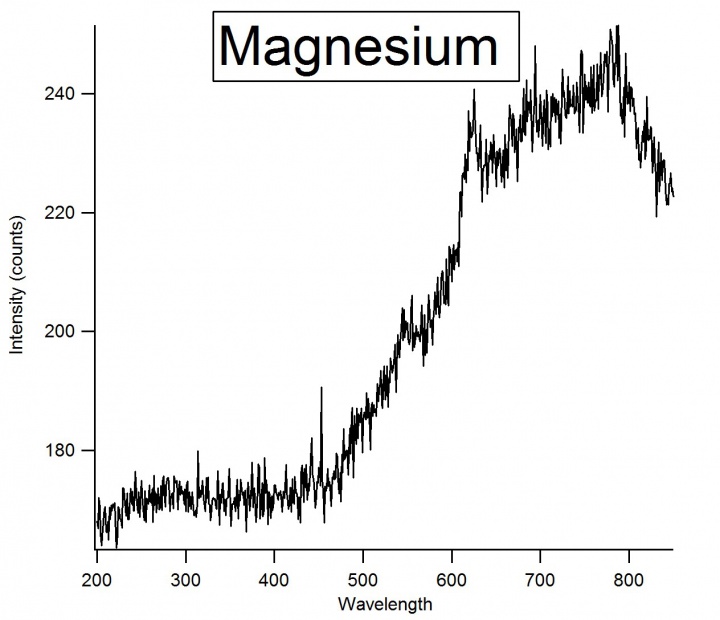
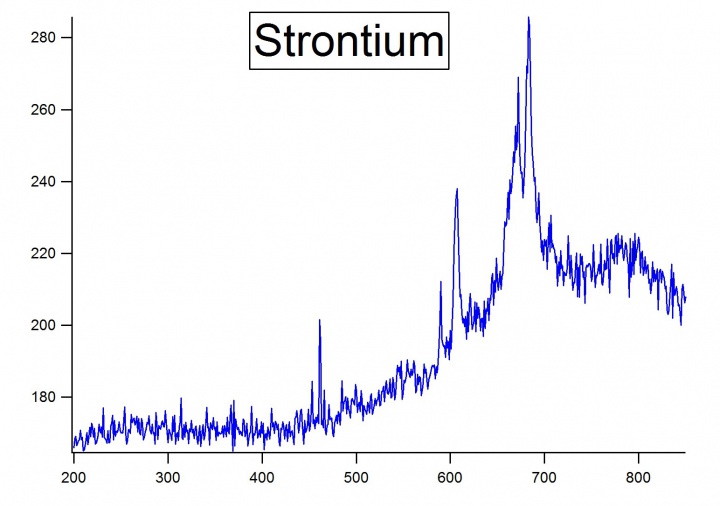
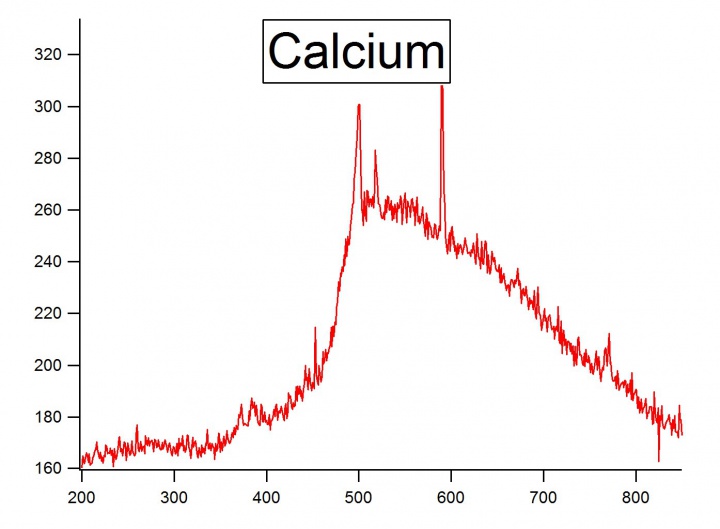
The instrument used for this experiment was the Ocean Optics Red Tide USB650 UV. Three metals were chosen for the experiment: Magnesium, Strontium, and Calcium. A magnesium ribbon, strontium, and calcium bits were used. The metals emission spectra were found by taking a piece of the metal and heating it up with a hand-held torch. First, the torch would heat the metal until the metal sparked. Once sparked it would emit a light. The length of emission of the light would depend on the metal and the amount. Once the metal emitted light, the distance from the metal to the detector had to be adjusted to get within a reasonable intensity. | The instrument used for this experiment was the Ocean Optics Red Tide USB650 UV. Three metals were chosen for the experiment: Magnesium, Strontium, and Calcium. A magnesium ribbon, strontium, and calcium bits were used. The metals emission spectra were found by taking a piece of the metal and heating it up with a hand-held torch. First, the torch would heat the metal until the metal sparked. Once sparked it would emit a light. The length of emission of the light would depend on the metal and the amount. Once the metal emitted light, the distance from the metal to the detector had to be adjusted to get within a reasonable intensity. | ||
[[File:Layout0.jpg|720px|thumb|left|Emission spectra of Magnesium Ribbon]] | [[File:Layout0.jpg|720px|thumb|left|Emission spectra of Magnesium Ribbon]] | ||
| + | [[File:Strontium.jpg|720px|thumb|left|Emission spectra of Strontium]] | ||
| + | [[File:Calcium.jpg|720px|thumb|left|Emission spectra of Calcium]] | ||
Latest revision as of 03:32, 2 February 2017
The instrument used for this experiment was the Ocean Optics Red Tide USB650 UV. Three metals were chosen for the experiment: Magnesium, Strontium, and Calcium. A magnesium ribbon, strontium, and calcium bits were used. The metals emission spectra were found by taking a piece of the metal and heating it up with a hand-held torch. First, the torch would heat the metal until the metal sparked. Once sparked it would emit a light. The length of emission of the light would depend on the metal and the amount. Once the metal emitted light, the distance from the metal to the detector had to be adjusted to get within a reasonable intensity.