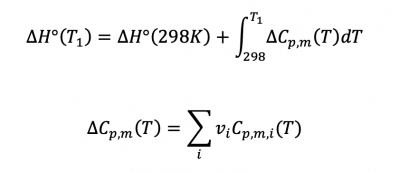Difference between revisions of "PChem312 f20 w11"
Jump to navigation
Jump to search
| (9 intermediate revisions by the same user not shown) | |||
| Line 6: | Line 6: | ||
'''Announcement''': Exam 2, Thursday, Oct 29th (Ch 3 and 4) | '''Announcement''': Exam 2, Thursday, Oct 29th (Ch 3 and 4) | ||
====Sec 4.4==== | ====Sec 4.4==== | ||
| − | + | Temperature dependence of reaction enthalpies... | |
| + | |||
| + | [[File:Screen Shot 2020-10-26 at 7.56.09 AM.png|400px]] | ||
| + | |||
| + | This problem involves 2 parts: | ||
| + | :1) ΔH°(298K) can be calculated via heats of formation (ΔH°<sub>f</sub>) | ||
| + | :2) the second part requires that you look up the C<sub>p,m</sub> values from table 2.5... | ||
| + | |||
| + | Study Example Problem 4.2 for calculation details... | ||
| + | :ΔH°(298K) = -92.3 kJ/mol HCl | ||
| + | : temp correction = -2.8 kJ/mol HCl | ||
| + | :ΔH°(298K) = -95.1 kJ/mol HCl | ||
| + | :'''''Explanation: this difference is mainly due to the change in the heat capacity of Cl<sub>2</sub> gas; the product HCl has a lower heat capacity than Cl<sub>2</sub>...so heat produced from the reaction cannot be contained in the products hence it is given off...ie. higher ΔH°(1450K).''''' | ||
| + | |||
| + | :[[File:ExProb4_2.png|600px]] | ||
| + | |||
| + | ====Additional worksheet==== | ||
| + | :[[Media:Self_heating_can.pdf|Self-Heating Can]] | ||
===Weds, Oct 28, 2020=== | ===Weds, Oct 28, 2020=== | ||
| − | + | ====Sec. 4.6 (supplental)==== | |
| + | Differential Scanning Calorimetry (DCS) | ||
===Thurs, Oct 29th, 2020=== | ===Thurs, Oct 29th, 2020=== | ||
Latest revision as of 15:31, 29 October 2020
Monday, Oct 26, 2020
Announcement: Exam 2, Thursday, Oct 29th (Ch 3 and 4)
Sec 4.4
Temperature dependence of reaction enthalpies...
This problem involves 2 parts:
- 1) ΔH°(298K) can be calculated via heats of formation (ΔH°f)
- 2) the second part requires that you look up the Cp,m values from table 2.5...
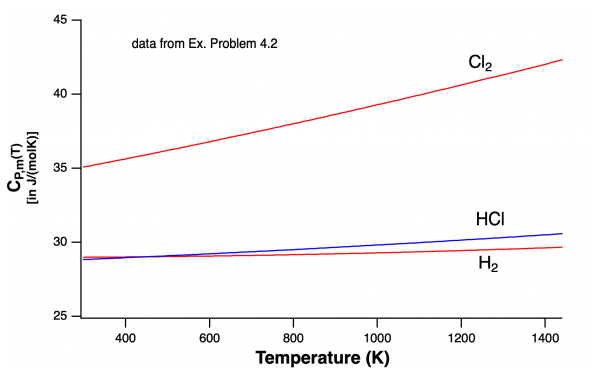
Study Example Problem 4.2 for calculation details...
- ΔH°(298K) = -92.3 kJ/mol HCl
- temp correction = -2.8 kJ/mol HCl
- ΔH°(298K) = -95.1 kJ/mol HCl
- Explanation: this difference is mainly due to the change in the heat capacity of Cl2 gas; the product HCl has a lower heat capacity than Cl2...so heat produced from the reaction cannot be contained in the products hence it is given off...ie. higher ΔH°(1450K).
Additional worksheet
Weds, Oct 28, 2020
Sec. 4.6 (supplental)
Differential Scanning Calorimetry (DCS)
Thurs, Oct 29th, 2020
- Exam 2, Ch 3 and 4.
Friday, Oct 30th, 2020
- no class