Difference between revisions of "Hannah"
Jump to navigation
Jump to search
(→WebMO) |
|||
| Line 25: | Line 25: | ||
:-All calculations were done using HF/6-31G(d) | :-All calculations were done using HF/6-31G(d) | ||
| − | =Keto Acetylacetone= | + | ==Keto Acetylacetone== |
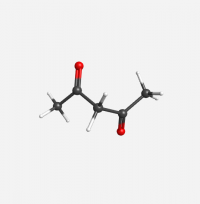
[[File:molecule (2).png|200px]] | [[File:molecule (2).png|200px]] | ||
Revision as of 21:23, 15 April 2021
Introduction
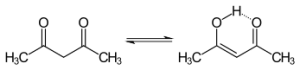
Tautomers of Acetylacetone (C5H8O2)
- -Keto = ketone
- -Enol = alcohol
- -may also be referred to as an alkenol
- -Usually the keto tautomer is favored
Why Tautomerization Matters
- -Different solvents may change the equilibrium of the keto- and enol- forms
- -Tautomers in DNA replication have been hypothesized to contribute to mutagenic mispairings
- -"whereas in RNA, minor tautomeric forms have been proposed to enhance the structural and functional diversity of RNA enzymes and aptamers" (https://rnajournal.cshlp.org/content/21/1/1.full.html)
WebMO
- -All calculations were done using HF/6-31G(d)
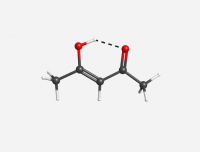
Keto Acetylacetone
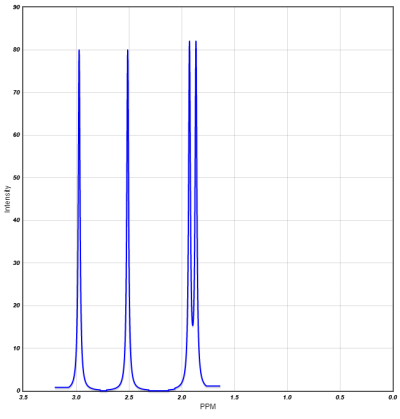
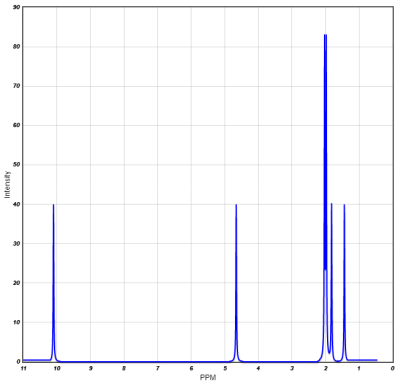
Proton NMR shifts:File:NMR-Chemical-Shift-PPM-Range-and-Values-Table-.png
Enol Acetylacetone
Proton NMR shifts:File:NMR-Chemical-Shift-PPM-Range-and-Values-Table-.png
N.B. Hydrogen bonding deshields the proton, further increasing the chemical shift