Difference between revisions of "Energy Barrier Analysis of the Middle C-C Bond in P3HT via Different Molecular Modeling Methods"
Jump to navigation
Jump to search
| Line 9: | Line 9: | ||
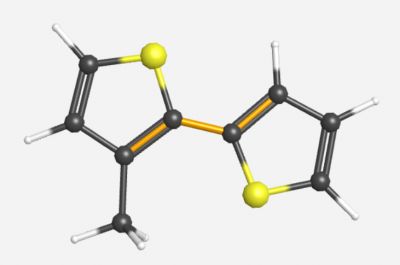
==Middle Dihedral Angle with One Methyl Substituent== | ==Middle Dihedral Angle with One Methyl Substituent== | ||
| + | |||
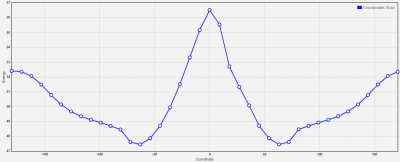
Symmetry: C<sub>S</sub> | Symmetry: C<sub>S</sub> | ||
| + | |||
| + | [[File:Screenshot 2021-04-08 200747.png|400px]] | ||
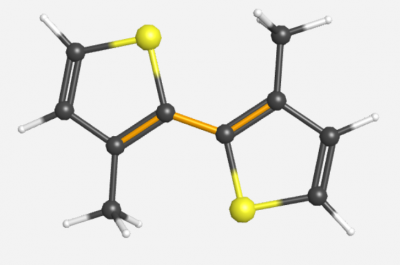
==Middle Dihedral Angle with Two Methyl Substituents== | ==Middle Dihedral Angle with Two Methyl Substituents== | ||
Revision as of 01:13, 9 April 2021
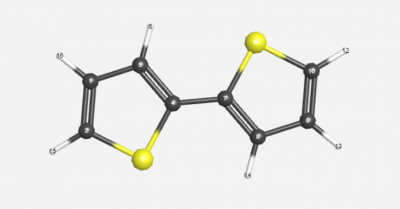
Middle Dihedral Angle with No Substituents
The molecule above was used for analysis. For the first molecule, the hydrocarbon substituents were removed.
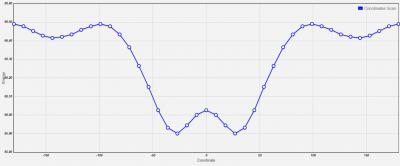
This calculation took 40.6 seconds to complete.
Middle Dihedral Angle with One Methyl Substituent
Symmetry: CS
Middle Dihedral Angle with Two Methyl Substituents
Symmetry: C2h