Difference between revisions of "PChem312 f20 w5"
Jump to navigation
Jump to search
| Line 47: | Line 47: | ||
:- using Igor ([https://www.wavemetrics.com/downloads/current/Igor%20Pro%208 Wavemetrics Igor 8.0 download]/[[File:Igor_8.jpg|50px]]) | :- using Igor ([https://www.wavemetrics.com/downloads/current/Igor%20Pro%208 Wavemetrics Igor 8.0 download]/[[File:Igor_8.jpg|50px]]) | ||
| − | 1) Cowboy Coffee worksheet ([[Media:cowboy coffee.pdf|here]]) | + | 1) Thermal imaging camera - insulated mugs |
| + | 2) Cowboy Coffee worksheet ([[Media:cowboy coffee.pdf|here]]) | ||
===Friday, Sept 18, 2020=== | ===Friday, Sept 18, 2020=== | ||
Revision as of 17:30, 17 September 2020
Monday, Sept 14, 2020
Announcement: Exam I (Ch 1 and 2), Thursday Sept 24th 2-6 pm
- - books allowed
- - notes allowed
- - computers required (computers in CSB 377 will be available)
Section 2.10
Read/skip
Section 2.11: Heat Capacity
- - CV,m - molar heat capacity at constant volume.
- - CP,m - molar heat capacity at constant pressure.
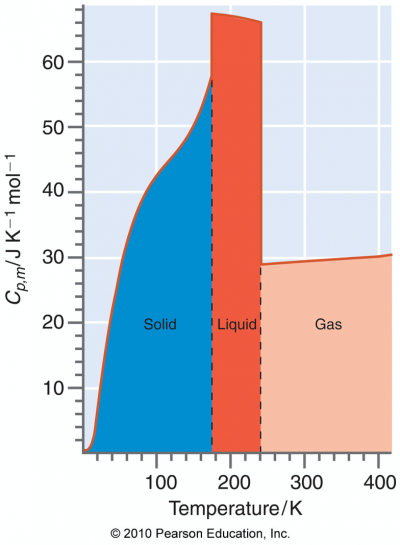
- heat capacities are usually temperature dependent, so CV,m(T) and CP,m(T); more later in Ch 3.
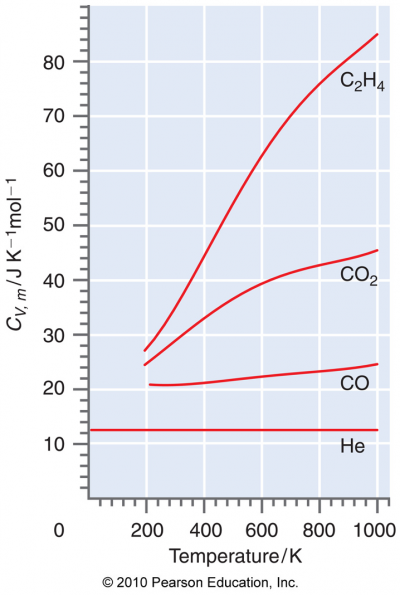
- - See Table 2.5: Molar Heat Capacity, CP,m(T), of Gases (298-800K), page 629.
Wednesday, Sept 16, 2020
Sec 2.11: continued
Relationship between CV,m and CP,m...
- CP - CV = nR
- CP,m - CV,m = R
Sec 2.12/2.13: FINALLY...Calculating ΔU...and more!
ΔU = q + w <-- 1st law of thermodynamics (mathematical form)
- Case 1...
- Consider a constant volume process where no expansion work (PV work) can be done...reminder for PV work, w = -PΔV...if constant volume (ΔV=0), then no expansion work...and then ΔU = qV, where we add the subscript "V" on q to indicate constant volume.
- ΔU = qV
- (we will work a few problems like this soon)
- Note: it would be nice if there was an equivalent for constant pressure (the much more common why of doing experiments...
- See "derivation" of ΔH (enthalpy - the second thermodynamic state function) (page 50-51).
- ΔH = qP
- Consider a constant volume process where no expansion work (PV work) can be done...reminder for PV work, w = -PΔV...if constant volume (ΔV=0), then no expansion work...and then ΔU = qV, where we add the subscript "V" on q to indicate constant volume.
Thursday, Sept 17, 2020
0) Heat Capacity Plot (use data from Heating/Cooling worksheet)
- - using Excel
- - using Igor (Wavemetrics Igor 8.0 download/
 )
)
1) Thermal imaging camera - insulated mugs 2) Cowboy Coffee worksheet (here)