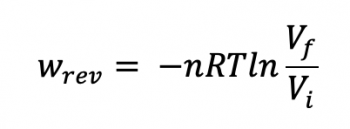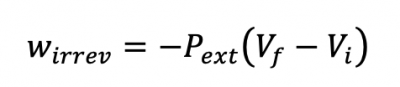Difference between revisions of "PChem312 f20 w4"
Jump to navigation
Jump to search
| Line 24: | Line 24: | ||
Sorry...still drawing a bunch of diagrams to help clarify the processes...i will send email when complete. | Sorry...still drawing a bunch of diagrams to help clarify the processes...i will send email when complete. | ||
| + | =====Irreversible Work===== | ||
The following shows a ''PV diagram'', also known as an '''indicator diagram'''. The center figure is Fig. 2.7 in your text. The outside "cylinders" are my annotation to clarify the diagram. I have separated the "expansion" (irreversible, isothermal) work from the "compression" (irreversible isothermal) work. | The following shows a ''PV diagram'', also known as an '''indicator diagram'''. The center figure is Fig. 2.7 in your text. The outside "cylinders" are my annotation to clarify the diagram. I have separated the "expansion" (irreversible, isothermal) work from the "compression" (irreversible isothermal) work. | ||
| Line 37: | Line 38: | ||
::Starting at an initial state (P<sub>2</sub>, V<sub>2</sub>; green dot), when we add the weight quickly, but do not allow the system to change volume, we find ourselves in a temporary/fictitious/non-equilibrium state (P<sub>1</sub>, V<sub>2</sub>; pink dot). The irreversible work is then done under P<sub>1</sub> (P<sub>1</sub> > P<sub>2</sub>) ending in the final state (P<sub>1</sub>, V<sub>1</sub>; yellow dot). Work was done '''''on the system''''' hence has a positive value. | ::Starting at an initial state (P<sub>2</sub>, V<sub>2</sub>; green dot), when we add the weight quickly, but do not allow the system to change volume, we find ourselves in a temporary/fictitious/non-equilibrium state (P<sub>1</sub>, V<sub>2</sub>; pink dot). The irreversible work is then done under P<sub>1</sub> (P<sub>1</sub> > P<sub>2</sub>) ending in the final state (P<sub>1</sub>, V<sub>1</sub>; yellow dot). Work was done '''''on the system''''' hence has a positive value. | ||
| + | |||
| + | =====Reversible Work===== | ||
===Thursday, Sept 10, 2020=== | ===Thursday, Sept 10, 2020=== | ||
Revision as of 15:42, 9 September 2020
Monday, Sept 7, 2020
Review: The first Law of thermodynamics: ΔU = q + w
Sec 2.5: Reversible Work, continued
- Example Problem 2.1-like
Sec 2.6: Irreversible Work, continued
- Example Problem 2.2-like
Sec 2.7: Other Work
- focus on electrical work, Example Prob 2.3c
Sec 2.8: State Functions and Path Functions
READ
Wednesday, Sept 9, 2020
Sec 2.9: Comparing Work/Indicator Diagrams
Sorry...still drawing a bunch of diagrams to help clarify the processes...i will send email when complete.
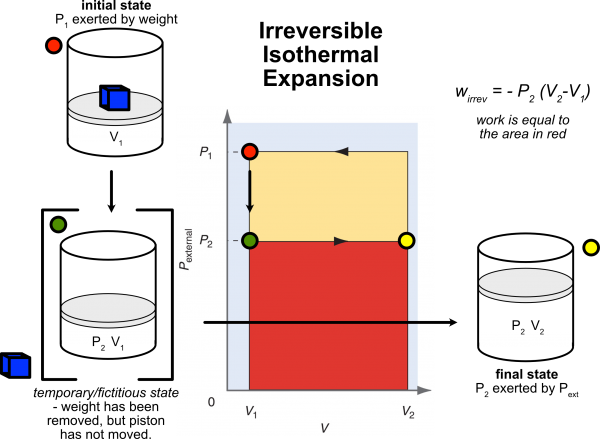
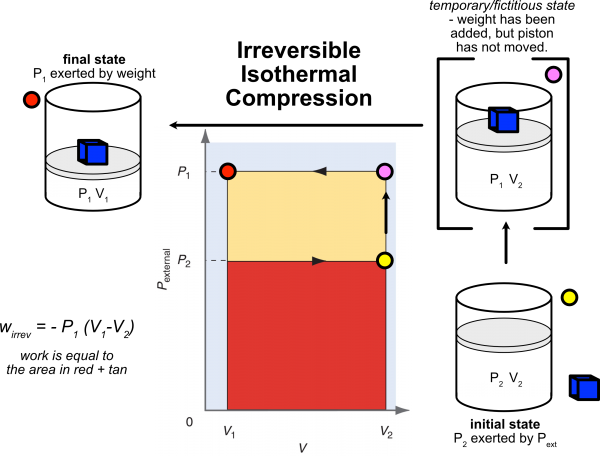
Irreversible Work
The following shows a PV diagram, also known as an indicator diagram. The center figure is Fig. 2.7 in your text. The outside "cylinders" are my annotation to clarify the diagram. I have separated the "expansion" (irreversible, isothermal) work from the "compression" (irreversible isothermal) work.
- Starting at an initial state (P1, V1; red dot), when we remove the weight quickly, but do not allow the system to change volume, we find ourselves in a temporary/fictitious/non-equilibrium state (P2, V1; green dot). The irreversible work is then done under P2 (P2 < P1) ending in the final state (P2, V2; yellow dot). Work was done by the system hence has a negative value.
- Starting at an initial state (P2, V2; green dot), when we add the weight quickly, but do not allow the system to change volume, we find ourselves in a temporary/fictitious/non-equilibrium state (P1, V2; pink dot). The irreversible work is then done under P1 (P1 > P2) ending in the final state (P1, V1; yellow dot). Work was done on the system hence has a positive value.
Reversible Work
Thursday, Sept 10, 2020
- - Thermometer (cont.)
- Fill thermometer
- Calibrate thermometer
- Seal thermometer