Difference between revisions of "Ch7 lec 2"
| Line 10: | Line 10: | ||
{| | {| | ||
|[[File:RBG-LED.jpg|400px]]||[[File:RGB_LED_Spectrum2.png|450px]] | |[[File:RBG-LED.jpg|400px]]||[[File:RGB_LED_Spectrum2.png|450px]] | ||
| + | |} | ||
| + | |||
| + | ===Problem very similar to #2 from lab=== | ||
| + | Convert the following table: | ||
| + | {| border=1 | ||
| + | | Wavelength (nm)|| Wavelength (m)||Frequency (1/s)||Energy (J) | ||
| + | |- | ||
| + | |650|| || || | ||
| + | |- | ||
| + | |570|| || || | ||
| + | |- | ||
| + | |480|| || || | ||
|} | |} | ||
Revision as of 11:59, 22 April 2020
(4/21/20, bes)
Additional Light Lab Info
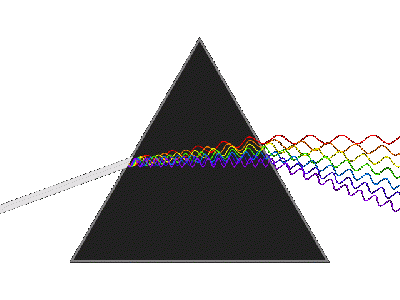
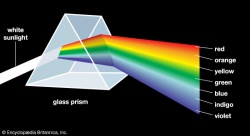
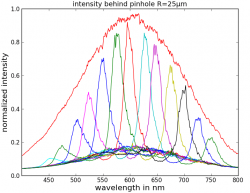
In our lab discussion i did not introduce you to the "photodiode array" (PDA) <-- not "public display of affection" --> instrument. A PDA is very similar to an electronic version of a "glass prism." A prism separates the wavelengths of light into the individual color components; when a rainbow is observed, the tiny water droplets are acting as a prism. If we were to direct "white" light into the PDA (which contains a prims and more), then you could measure/collect a "spectrum" similar to the "broad," featureless spectrum (red) on the far right figure...note the wavelengths are in nanometers (nm). This figure is complicated by the fact that these authors used a technique to separate the light prior to it entering the PDA, so that is why there are all of the other "narrow" spectra also shown.
 |
 |
|
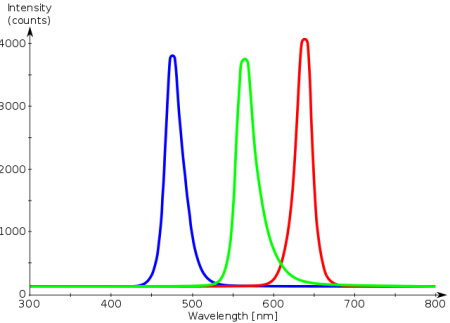
Sorry, i know this is a little confusing...hang in there...here is another example...I think you should be familiar with LEDs (light emitting diodes)...they come in different colors. If we were to meet in person for the "Light Lab", then i would have shown you the PDA spectrum of the LEDs. In the figure below, the red, green, and blue LEDs when shown into the PDA would result in a spectrum with a peak at 650 nm - red, 570 nm - green, and 480 nm - blue (pls note, there are 3 separate spectra on this one graph).
 |
|
Problem very similar to #2 from lab
Convert the following table:
| Wavelength (nm) | Wavelength (m) | Frequency (1/s) | Energy (J) |
| 650 | |||
| 570 | |||
| 480 |