Difference between revisions of "ESR Lab Activity"
Jump to navigation
Jump to search
(→WinSim) |
|||
| Line 81: | Line 81: | ||
||HF/6311G+(2p,d) | ||HF/6311G+(2p,d) | ||
||B3LYP/6-31G | ||B3LYP/6-31G | ||
| − | || | + | ||B3LYP/3-21G |
||B3LYP/6311G+(2p,d) | ||B3LYP/6311G+(2p,d) | ||
|| | || | ||
| Line 92: | Line 92: | ||
|| | || | ||
||1.10213 | ||1.10213 | ||
| − | || | + | ||0.85863 |
||0.52092 | ||0.52092 | ||
|| | || | ||
| Line 103: | Line 103: | ||
|| | || | ||
||-2.73487 | ||-2.73487 | ||
| − | || | + | ||-2.67806 |
||-2.55611 | ||-2.55611 | ||
|| | || | ||
| Line 114: | Line 114: | ||
|| | || | ||
||-4.10834 | ||-4.10834 | ||
| − | || | + | ||-2.68141 |
||-2.55653 | ||-2.55653 | ||
|| | || | ||
| Line 125: | Line 125: | ||
|| | || | ||
||1.27101 | ||1.27101 | ||
| − | || | + | ||0.85927 |
||0.52095 | ||0.52095 | ||
|| | || | ||
| Line 136: | Line 136: | ||
|| | || | ||
||1.27509 | ||1.27509 | ||
| − | || | + | ||0.85858 |
||0.52091 | ||0.52091 | ||
|| | || | ||
| Line 147: | Line 147: | ||
|| | || | ||
||-4.09995 | ||-4.09995 | ||
| − | || | + | ||-2.67694 |
||-2.55567 | ||-2.55567 | ||
|| | || | ||
| Line 158: | Line 158: | ||
|| | || | ||
||-2.74025 | ||-2.74025 | ||
| − | || | + | ||-2.68052 |
||-2.55603 | ||-2.55603 | ||
|| | || | ||
| Line 169: | Line 169: | ||
|| | || | ||
||1.10424 | ||1.10424 | ||
| − | || | + | ||-0.58918 |
||0.52091 | ||0.52091 | ||
|| | || | ||
| Line 180: | Line 180: | ||
|| | || | ||
||3.46774 | ||3.46774 | ||
| − | || | + | ||-0.22807 |
||-0.28092 | ||-0.28092 | ||
|| | || | ||
| Line 191: | Line 191: | ||
|| | || | ||
||3.27784 | ||3.27784 | ||
| − | || | + | ||-0.22870 |
||-0.28078 | ||-0.28078 | ||
|| | || | ||
Revision as of 20:34, 28 April 2016
Introduction
We are going to collect an ESR spectrum from a series of substituted hydroquinones.
| Name | SDS | Molar Mass | Sigma Product Number | Cost |
| A = Hydroquinone (aka. 1,4-benzohydroquinone) | SDS | 110.11 g/mol | H9003 | 22.90 / 100g |
| B = Methylhydroquinone | SDS | 124.14 g/mol | 112968 | 59.20 / 250g |
| C = 2,3-dimethylhydroquinone | SDS | 138.16 g/mol | 300756 | 118.00 / 5g |
Experimental
Beaker Method
- Solution A: 1 M NaOH; 1 gram NaOH (39.997 g/mol) into 25 mL EtOH.
- Solution B: 1 M hydroquinone solution.
- Procedure: To 2 ml of 1 M hydroquinone solution add 2-3 drops of solution A. A color change will indicate the reaction has occurred. Quickly transfer colored sample to ESR sample tube, place in ESR spectrometer, tune, and collected data.
Flow Method
- Solution A: 0.05 M NaOH; 0.05 grams NaOH (39.997 g/mol) into 25 ml EtOH.
- Solution B: 1 M hydroquinone solution.
- Procedure: Prepare two 60 ml syringes, 1 with solution A and 1 with solution B.
- Degas the syringe that contains solution B
- Attach to double syringe drive
- Turn on double syringe drive that is attached to ESR
- Collect data
Double Syringe Drive
Results
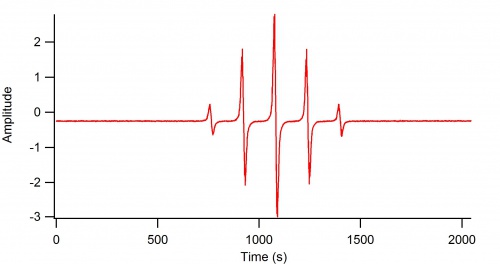
1-electron Oxidation of Hydroquinone
This is the EPR spectrum of the "hydro-semiquinone" (aka. 1,4-benzosemiquinone)
EPR Parameters: 9.4 GHz, 3360 G Center field, 15 G sweep width. [Above data needs the x-axis changed over to magnetic field in G]
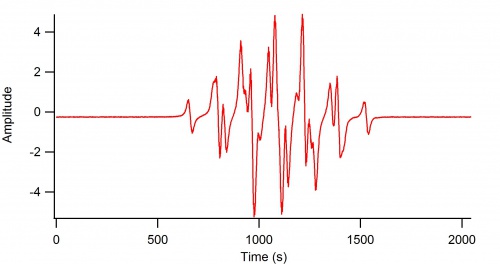
1-electron Oxidation of Methylhydroquinone
This is the EPR spectrum of the "methyl-hydro-semiquinone" (aka. methyl-semiquinone)
EPR Parameters: 9.4 GHz, 3360 G Center field, 15 G sweep width. [Above data needs the x-axis changed over to magnetic field in G]
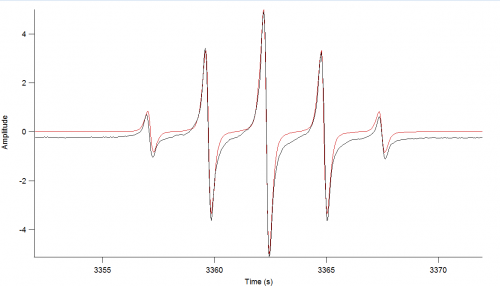
Analysis
WinSim
Gaussian Calculations
| Atom | HF/3-21G | HF/6-31G | HF/6311G+(2p,d) | B3LYP/6-31G | B3LYP/3-21G | B3LYP/6311G+(2p,d) | |||
| 8 | 12.605 | 1.10213 | 0.85863 | 0.52092 | |||||
| 9 | -14.289 | -2.73487 | -2.67806 | -2.55611 | |||||
| 11 | -14.289 | -4.10834 | -2.68141 | -2.55653 | |||||
| 12 | 12.604 | 1.27101 | 0.85927 | 0.52095 | |||||
| 19 | 12.604 | 1.27509 | 0.85858 | 0.52091 | |||||
| 20 | -14.289 | -4.09995 | -2.67694 | -2.55567 | |||||
| 22 | -14.289 | -2.74025 | -2.68052 | -2.55603 | |||||
| 23 | 12.604 | 1.10424 | -0.58918 | 0.52091 | |||||
| 24 | 0.661 | 3.46774 | -0.22807 | -0.28092 | |||||
| 25 | 0.660 | 3.27784 | -0.22870 | -0.28078 |